Have a language expert improve your writing
Run a free plagiarism check in 10 minutes, generate accurate citations for free.
- Knowledge Base
- Starting the research process
- Writing Strong Research Questions | Criteria & Examples

Writing Strong Research Questions | Criteria & Examples
Published on October 26, 2022 by Shona McCombes . Revised on November 21, 2023.
A research question pinpoints exactly what you want to find out in your work. A good research question is essential to guide your research paper , dissertation , or thesis .
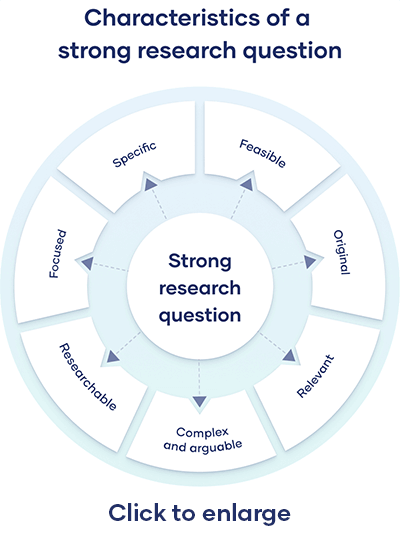
All research questions should be:
- Focused on a single problem or issue
- Researchable using primary and/or secondary sources
- Feasible to answer within the timeframe and practical constraints
- Specific enough to answer thoroughly
- Complex enough to develop the answer over the space of a paper or thesis
- Relevant to your field of study and/or society more broadly
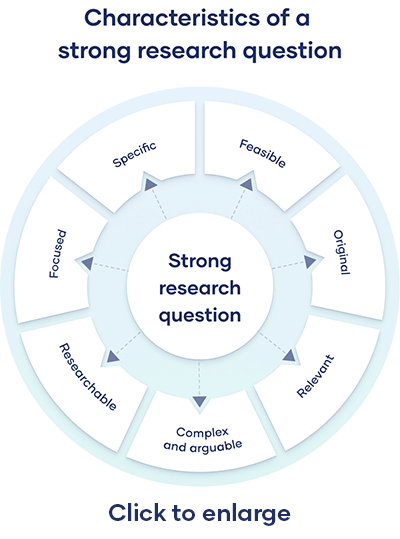
Table of contents
How to write a research question, what makes a strong research question, using sub-questions to strengthen your main research question, research questions quiz, other interesting articles, frequently asked questions about research questions.
You can follow these steps to develop a strong research question:
- Choose your topic
- Do some preliminary reading about the current state of the field
- Narrow your focus to a specific niche
- Identify the research problem that you will address
The way you frame your question depends on what your research aims to achieve. The table below shows some examples of how you might formulate questions for different purposes.
| Research question formulations | |
|---|---|
| Describing and exploring | |
| Explaining and testing | |
| Evaluating and acting | is X |
Using your research problem to develop your research question
| Example research problem | Example research question(s) |
|---|---|
| Teachers at the school do not have the skills to recognize or properly guide gifted children in the classroom. | What practical techniques can teachers use to better identify and guide gifted children? |
| Young people increasingly engage in the “gig economy,” rather than traditional full-time employment. However, it is unclear why they choose to do so. | What are the main factors influencing young people’s decisions to engage in the gig economy? |
Note that while most research questions can be answered with various types of research , the way you frame your question should help determine your choices.
Prevent plagiarism. Run a free check.
Research questions anchor your whole project, so it’s important to spend some time refining them. The criteria below can help you evaluate the strength of your research question.
Focused and researchable
| Criteria | Explanation |
|---|---|
| Focused on a single topic | Your central research question should work together with your research problem to keep your work focused. If you have multiple questions, they should all clearly tie back to your central aim. |
| Answerable using | Your question must be answerable using and/or , or by reading scholarly sources on the to develop your argument. If such data is impossible to access, you likely need to rethink your question. |
| Not based on value judgements | Avoid subjective words like , , and . These do not give clear criteria for answering the question. |
Feasible and specific
| Criteria | Explanation |
|---|---|
| Answerable within practical constraints | Make sure you have enough time and resources to do all research required to answer your question. If it seems you will not be able to gain access to the data you need, consider narrowing down your question to be more specific. |
| Uses specific, well-defined concepts | All the terms you use in the research question should have clear meanings. Avoid vague language, jargon, and too-broad ideas. |
| Does not demand a conclusive solution, policy, or course of action | Research is about informing, not instructing. Even if your project is focused on a practical problem, it should aim to improve understanding rather than demand a ready-made solution. If ready-made solutions are necessary, consider conducting instead. Action research is a research method that aims to simultaneously investigate an issue as it is solved. In other words, as its name suggests, action research conducts research and takes action at the same time. |
Complex and arguable
| Criteria | Explanation |
|---|---|
| Cannot be answered with or | Closed-ended, / questions are too simple to work as good research questions—they don’t provide enough for robust investigation and discussion. |
| Cannot be answered with easily-found facts | If you can answer the question through a single Google search, book, or article, it is probably not complex enough. A good research question requires original data, synthesis of multiple sources, and original interpretation and argumentation prior to providing an answer. |
Relevant and original
| Criteria | Explanation |
|---|---|
| Addresses a relevant problem | Your research question should be developed based on initial reading around your . It should focus on addressing a problem or gap in the existing knowledge in your field or discipline. |
| Contributes to a timely social or academic debate | The question should aim to contribute to an existing and current debate in your field or in society at large. It should produce knowledge that future researchers or practitioners can later build on. |
| Has not already been answered | You don’t have to ask something that nobody has ever thought of before, but your question should have some aspect of originality. For example, you can focus on a specific location, or explore a new angle. |
Chances are that your main research question likely can’t be answered all at once. That’s why sub-questions are important: they allow you to answer your main question in a step-by-step manner.
Good sub-questions should be:
- Less complex than the main question
- Focused only on 1 type of research
- Presented in a logical order
Here are a few examples of descriptive and framing questions:
- Descriptive: According to current government arguments, how should a European bank tax be implemented?
- Descriptive: Which countries have a bank tax/levy on financial transactions?
- Framing: How should a bank tax/levy on financial transactions look at a European level?
Keep in mind that sub-questions are by no means mandatory. They should only be asked if you need the findings to answer your main question. If your main question is simple enough to stand on its own, it’s okay to skip the sub-question part. As a rule of thumb, the more complex your subject, the more sub-questions you’ll need.
Try to limit yourself to 4 or 5 sub-questions, maximum. If you feel you need more than this, it may be indication that your main research question is not sufficiently specific. In this case, it’s is better to revisit your problem statement and try to tighten your main question up.
Here's why students love Scribbr's proofreading services
Discover proofreading & editing
If you want to know more about the research process , methodology , research bias , or statistics , make sure to check out some of our other articles with explanations and examples.
Methodology
- Sampling methods
- Simple random sampling
- Stratified sampling
- Cluster sampling
- Likert scales
- Reproducibility
Statistics
- Null hypothesis
- Statistical power
- Probability distribution
- Effect size
- Poisson distribution
Research bias
- Optimism bias
- Cognitive bias
- Implicit bias
- Hawthorne effect
- Anchoring bias
- Explicit bias
The way you present your research problem in your introduction varies depending on the nature of your research paper . A research paper that presents a sustained argument will usually encapsulate this argument in a thesis statement .
A research paper designed to present the results of empirical research tends to present a research question that it seeks to answer. It may also include a hypothesis —a prediction that will be confirmed or disproved by your research.
As you cannot possibly read every source related to your topic, it’s important to evaluate sources to assess their relevance. Use preliminary evaluation to determine whether a source is worth examining in more depth.
This involves:
- Reading abstracts , prefaces, introductions , and conclusions
- Looking at the table of contents to determine the scope of the work
- Consulting the index for key terms or the names of important scholars
A research hypothesis is your proposed answer to your research question. The research hypothesis usually includes an explanation (“ x affects y because …”).
A statistical hypothesis, on the other hand, is a mathematical statement about a population parameter. Statistical hypotheses always come in pairs: the null and alternative hypotheses . In a well-designed study , the statistical hypotheses correspond logically to the research hypothesis.
Formulating a main research question can be a difficult task. Overall, your question should contribute to solving the problem that you have defined in your problem statement .
However, it should also fulfill criteria in three main areas:
- Researchability
- Feasibility and specificity
- Relevance and originality
Cite this Scribbr article
If you want to cite this source, you can copy and paste the citation or click the “Cite this Scribbr article” button to automatically add the citation to our free Citation Generator.
McCombes, S. (2023, November 21). Writing Strong Research Questions | Criteria & Examples. Scribbr. Retrieved June 24, 2024, from https://www.scribbr.com/research-process/research-questions/
Is this article helpful?
Shona McCombes
Other students also liked, how to define a research problem | ideas & examples, how to write a problem statement | guide & examples, 10 research question examples to guide your research project, get unlimited documents corrected.
✔ Free APA citation check included ✔ Unlimited document corrections ✔ Specialized in correcting academic texts

Module 2: Formulating a Research Question and Searching for Sources
Formulating a Research Question
As noted in Module 1: Types of Reviews , conducting a “pre-search” is a crucial first step in devising the research question. A well-formulated research question informs the research process. It can focus your information needs (i.e. identify inclusion and exclusion criteria), help to identify key search concepts, and guide you in the direction of relevant resources.
Types of Research Questions
There are two general types of research questions: quantitative and qualitative .
Quantitative Research Questions
Types of quantitative questions can be categorized as explanatory (i.e., relationship-based), descriptive, or comparative.
- Explanatory questions aim to discover cause-and-effect relationships by comparing two or more variables, individuals or groups based on differing outcomes.
- Descriptive questions will often quantify a single variable but may include multiple variables within a question. They typically ask for measurements, and can begin with: “how much”, “what percentage”, “how frequently”, or “how many”.
- Comparative questions are designed to identify the “difference between” a dependent variable and two or more groups. These questions tend to begin with “what is the difference” or “what are the differences”.
Qualitative Research Questions
Qualitative questions aim to discover meaning or gain an understanding of a phenomenon. They ask questions that cannot be measured with specific numbers and statistics. Qualitative research questions often contain words like "lived experience" , “personal experience”, “understanding”, “meaning”, and “stories”.
A Comparison of Quantitative and Qualitative Questions
So how do quantitative and qualitative research questions differ when you are conducting a search? In Table 2.1 below, we provide some examples of research topics. Each topic can either be used for a quantitative or qualitative research question. For each question, the category of research question is clarified.
| How does experiencing housing-insecurity in childhood impact a child’s relationships in adulthood? | What are the lived experiences of children experiencing housing-insecurity while attending elementary School? | |
| What percentage of nurses experience violence in the workplace on night shifts? | How do nurses conceptualize their role in mitigating workplace violence? | |
| What are the differences in perceptions towards accessing mental health services between immigrants and their Canadian-born children? | How do newly arrived Canadians experience seeking help for mental health concerns? |
Key Takeaways
Regardless of the type of question being asked, a good research question cannot be answered with a simple yes or no (as demonstrated by the sample questions in Table 2.1).
Quantitative data can be counted, measured, and expressed using numbers.
Qualitative research relies on data obtained by the researcher from first-hand observation, interviews, questionnaires (on which participants write descriptively), focus groups, participant-observation, recordings made in natural settings, documents, and artifacts. The data are generally nonnumerical.
Personal knowledge about the world that has been gained through first-hand involvement in everyday events.
Advanced Research Skills: Conducting Literature and Systematic Reviews Copyright © 2021 by Kelly Dermody; Cecile Farnum; Daniel Jakubek; Jo-Anne Petropoulos; Jane Schmidt; and Reece Steinberg is licensed under a Creative Commons Attribution 4.0 International License , except where otherwise noted.
Share This Book
An official website of the United States government
The .gov means it’s official. Federal government websites often end in .gov or .mil. Before sharing sensitive information, make sure you’re on a federal government site.
The site is secure. The https:// ensures that you are connecting to the official website and that any information you provide is encrypted and transmitted securely.
- Publications
- Account settings
Preview improvements coming to the PMC website in October 2024. Learn More or Try it out now .
- Advanced Search
- Journal List
- Indian J Anaesth
- v.63(8); 2019 Aug
Formulating a good research question: Pearls and pitfalls
Wilson fandino.
Guys' and St Thomas' Hospital National Health Service Foundation Trust, London, United Kingdom
The process of formulating a good research question can be challenging and frustrating. While a comprehensive literature review is compulsory, the researcher usually encounters methodological difficulties in the conduct of the study, particularly if the primary study question has not been adequately selected in accordance with the clinical dilemma that needs to be addressed. Therefore, optimising time and resources before embarking in the design of a clinical protocol can make an impact on the final results of the research project. Researchers have developed effective ways to convey the message of how to build a good research question that can be easily recalled under the acronyms of PICOT (population, intervention, comparator, outcome, and time frame) and FINER (feasible, interesting, novel, ethical, and relevant). In line with these concepts, this article highlights the main issues faced by clinicians, when developing a research question.
INTRODUCTION
What is your research question? This is very often one of the first queries made by statisticians, when researchers come up with an interesting idea. In fact, the findings of a study may only acquire relevance if they provide an accurate and unbiased answer to a specific question,[ 1 , 2 ] and it has been suggested that up to one-third of the time spent in the whole process—from the conception of an idea to the publication of the manuscript—could be invested in finding the right primary study question.[ 3 ] Furthermore, selecting a good research question can be a time-consuming and challenging task: in one retrospective study, Mayo et al . reported that 3 out of 10 articles published would have needed a major rewording of the question.[ 1 ] This paper explores some recommendations to consider before starting any research project, and outlines the main difficulties faced by young and experienced clinicians, when it comes time to turn an exciting idea into a valuable and feasible research question.

OPTIMISATION OF TIME AND RESOURCES
Focusing on the primary research question.
The process of developing a new idea usually stems from a dilemma inherent to the clinical practice.[ 2 , 3 , 4 ] However, once the problem has been identified, it is tempting to formulate multiple research questions. Conducting a clinical trial with more than one primary study question would not be feasible. First, because each question may require a different research design, and second, because the necessary statistical power of the study would demand unaffordable sample sizes. It is the duty of editors and reviewers to make sure that authors clearly identify the primary research question, and as a consequence, studies approaching more than one primary research question may not be suitable for publication.
Working in the right environment
Teamwork is essential to find the appropriate research question. Working in the right environment will enable the investigator to interact with colleagues with different backgrounds, and create opportunities to exchange experiences in a collaborative way between clinicians and researchers. Likewise, it is of paramount importance to get involved colleagues with expertise in the field (lead clinicians, education supervisors, research mentors, department chairs, epidemiologists, biostatisticians, and ethical consultants, among others), and ask for their guidance.[ 5 , 6 , 7 , 8 ]
Evaluating the pertinence of the study
The researcher should wonder if, on the basis of the research question formulated, there is a need for a study to address the problem, as clinical research usually entails a large investment of resources and workforce involvement. Thus, if the answer to the posed clinical question seems to be evident before starting the study, investing in research to address the problem would become superfluous. For example, in a clinical trial, Herzog-Niescery et al . compared laryngeal masks with cuffed and uncuffed tracheal tubes, in the context of surgeons' exposure to sevoflurane, in infants undergoing adenoidectomy. However, it appears obvious that cuffed tracheal tubes are preferred to minimise surgeons' exposure to volatile gases, as authors concluded after recruiting 60 patients.[ 9 ]
Conducting a thorough literature review
Any research project requires the identification of at least one of three problems: the evidence is scarce, the existing literature yields conflicting results, or the results could be improved. Hence, a comprehensive review of the topic is imperative, as it allows the researcher to identify this gap in the literature, formulate a hypothesis and develop a research question.[ 2 ] To this end, it is crucial to be attentive to new ideas, keep the imagination roaming with reflective attitude, and remain sceptical to the new-gained information.[ 4 , 7 ]
Narrowing the research question
A broad research question may encompass an unaffordable extensive topic. For instance, do supraglottic devices provide similar conditions for the visualization of the glottis aperture in a German hospital? Such a general research question usually needs to be narrowed, not only by cutting away unnecessary components (a German hospital is irrelevant in this context), but also by defining a target population, a specific intervention, an alternative treatment or procedure to be compared with the intervention, a measurable primary outcome, and a time frame of the study. In contrast, an example of a good research question would be: among children younger than 1 year of age undergoing elective minor procedures, to what extent the insertion times are different, comparing the Supreme™ laryngeal mask airway (LMA) to Proseal™ LMA, when placed after reaching a BIS index <60?[ 10 ] In this example, the core ingredients of the research question can be easily identified as: children <1 year of age undergoing minor elective procedures, Supreme™ LMA, Proseal™ LMA and insertion times at anaesthetic induction when reaching a BIS index <60. These components are usually gathered in the literature under the acronym of PICOT (population, intervention, comparator, outcome and time frame, respectively).[ 1 , 3 , 5 ]
PICOT FRAMEWORK
Table 1 summarises the foremost questions likely to be addressed when working on PICOT frame.[ 1 , 6 , 8 ] These components are also applicable to observational studies, where the exposure takes place of the intervention.[ 1 , 11 ] Remarkably, if after browsing the title and the abstract of a paper, the reader is not able to clearly identify the PICOT parameters, and elucidate the question posed by the authors, there should be reasonable scepticism regarding the scientific rigor of the work.[ 12 , 13 ] All these elements are crucial in the design and methodology of a clinical trial, as they can affect the feasibility and reliability of results. Having formulated the primary study question in the context of the PICOT framework [ Table 1 ],[ 1 , 6 , 8 ] the researcher should be able to elucidate which design is most suitable for their work, determine what type of data needs to be collected, and write a structured introduction tailored to what they want to know, explicitly mentioning the primary study hypothesis, which should lead to formulate the main research question.[ 1 , 2 , 6 , 8 ]
Key questions to be answered when working with the PICOT framework (population, intervention, comparator, outcome, and time frame) in a clinical research design
| Component | Related questions |
|---|---|
| Population | -What is the target population? -Is the target population narrow or broad? -Is the target population vulnerable? -What are the eligibility criteria? -What is the most appropriate recruitment strategy? |
| Intervention | -What is the intervention? (treatment, diagnostic test, procedure) -Is there any standard of care for the intervention? -Is the intervention the most appropriate for the study design? -Is there a need for standardizing the intervention? -What are the potential side effects of the intervention? -Will potential side effects be recorded? -If there is no intervention, what is the exposure? |
| Comparator | -How has control intervention been chosen? -Are there any ethical concerns related to the use of placebo? -Has a sham intervention been considered? -Will statistical analyses be adjusted for multiple comparisons? |
| Outcome | -What is the primary outcome? -What are the secondary outcomes? -Are the outcomes exploratory, explanatory or confirmatory? -Have surrogate and clinical outcomes been considered? -Are the outcomes validated? -Have safety outcomes been considered? -How are the outcomes going to be measured? -Will the dependent and independent variables be numerical, categorical or ordinal? -Will be enough statistical power to measure secondary outcomes? |
| Time frame | -Is the study designed to be cross -sectional or longitudinal? -How long will the recruitment phase take? -What is the time frame for data collection? -Have frequency and duration of the intervention been specified? -How often will outcomes be measured? -Which strategy will be used to prevent/decrease dropouts? |
Occasionally, the intended population of the study needs to be modified, in order to overcome any potential ethical issues, and/or for the sake of convenience and feasibility of the project. Yet, the researcher must be aware that the external validity of the results may be compromised. As an illustration, in a randomised clinical trial, authors compared the ease of tracheal tube insertion between C-MAC video laryngoscope and direct laryngoscopy, in patients presenting to the emergency department with an indication of rapid sequence intubation. However, owing to the existence of ethical concerns, a substantial amount of patients requiring emergency tracheal intubation, including patients with major maxillofacial trauma and ongoing cardiopulmonary resuscitation, had to be excluded from the trial.[ 14 ] In fact, the design of prospective studies to explore this subset of patients can be challenging, not only because of ethical considerations, but because of the low incidence of these cases. In another study, Metterlein et al . compared the glottis visualisation among five different supraglottic airway devices, using fibreroptic-guided tracheal intubation in an adult population. Despite that the study was aimed to explore the ease of intubation in patients with anticipated difficult airway (thus requiring fibreoptic tracheal intubation), authors decided to enrol patients undergoing elective laser treatment for genital condylomas, as a strategy to hasten the recruitment process and optimise resources.[ 15 ]
Intervention
Anaesthetic interventions can be classified into pharmacological (experimental treatment) and nonpharmacological. Among nonpharmacological interventions, the most common include anaesthetic techniques, monitoring instruments and airway devices. For example, it would be appropriate to examine the ease of insertion of Supreme™ LMA, when compared with ProSeal™ LMA. Notwithstanding, a common mistake is the tendency to be focused on the data aimed to be collected (the “stated” objective), rather than the question that needs to be answered (the “latent” objective).[ 1 , 4 ] In one clinical trial, authors stated: “we compared the Supreme™ and ProSeal™ LMAs in infants by measuring their performance characteristics, including insertion features, ventilation parameters, induced changes in haemodynamics, and rates of postoperative complications”.[ 10 ] Here, the research question has been centered on the measurements (insertion characteristics, haemodynamic variables, LMA insertion characteristics, ventilation parameters) rather than the clinical problem that needs to be addressed (is Supreme™ LMA easier to insert than ProSeal™ LMA?).
Comparators in clinical research can also be pharmacological (e.g., gold standard or placebo) or nonpharmacological. Typically, not more than two comparator groups are included in a clinical trial. Multiple comparisons should be generally avoided, unless there is enough statistical power to address the end points of interest, and statistical analyses have been adjusted for multiple testing. For instance, in the aforementioned study of Metterlein et al .,[ 15 ] authors compared five supraglottic airway devices by recruiting only 10--12 participants per group. In spite of the authors' recommendation of using two supraglottic devices based on the results of the study, there was no mention of statistical adjustments for multiple comparisons, and given the small sample size, larger clinical trials will undoubtedly be needed to confirm or refute these findings.[ 15 ]
A clear formulation of the primary outcome results of vital importance in clinical research, as the primary statistical analyses, including the sample size calculation (and therefore, the estimation of the effect size and statistical power), will be derived from the main outcome of interest. While it is clear that using more than one primary outcome would not be appropriate, it would be equally inadequate to include multiple point measurements of the same variable as the primary outcome (e.g., visual analogue scale for pain at 1, 2, 6, and 12 h postoperatively).
Composite outcomes, in which multiple primary endpoints are combined, may make it difficult to draw any conclusions based on the study findings. For example, in a clinical trial, 200 children undergoing ophthalmic surgery were recruited to explore the incidence of respiratory adverse events, when comparing desflurane with sevoflurane, following the removal of flexible LMA during the emergence of the anaesthesia. The primary outcome was the number of respiratory events, including breath holding, coughing, secretions requiring suction, laryngospasm, bronchospasm, and mild desaturation.[ 16 ] Should authors had claimed a significant difference between these anaesthetic volatiles, it would have been important to elucidate whether those differences were due to serious adverse events, like laryngospasm or bronchospasm, or the results were explained by any of the other events (e.g., secretions requiring suction). While it is true that clinical trials evaluating the occurrence of adverse events like laryngospasm/bronchospasm,[ 16 , 17 ] or life-threating complications following a tracheal intubation (e.g., inadvertent oesophageal placement, dental damage or injury of the larynx/pharynx)[ 14 ] are almost invariably underpowered, because the incidence of such events is expected to be low, subjective outcomes like coughing or secretions requiring suction should be avoided, as they are highly dependent on the examiner's criteria.[ 16 ]
Secondary outcomes are useful to document potential side effects (e.g., gastric insufflation after placing a supraglottic device), and evaluate the adherence (say, airway leak pressure) and safety of the intervention (for instance, occurrence, or laryngospasm/bronchospasm).[ 17 ] Nevertheless, the problem of addressing multiple secondary outcomes without the adequate statistical power is habitual in medical literature. A good illustration of this issue can be found in a study evaluating the performance of two supraglottic devices in 50 anaesthetised infants and neonates, whereby authors could not draw any conclusions in regard to potential differences in the occurrence of complications, because the sample size calculated made the study underpowered to explore those differences.[ 17 ]
Among PICOT components, the time frame is the most likely to be omitted or inappropriate.[ 1 , 12 ] There are two key aspects of the time component that need to be clearly specified in the research question: the time of measuring the outcome variables (e.g. visual analogue scale for pain at 1, 2, 6, and 12 h postoperatively), and the duration of each measurement (when indicated). The omission of these details in the study protocol might lead to substantial differences in the methodology used. For instance, if a study is designed to compare the insertion times of three different supraglottic devices, and researchers do not specify the exact moment of LMA insertion in the clinical trial protocol (i.e., at the anaesthetic induction after reaching a BIS index < 60), placing an LMA with insufficient depth of anaesthesia would have compromised the internal validity of the results, because inserting a supraglottic device in those patients would have resulted in failed attempts and longer insertion times.[ 10 ]
FINER CRITERIA
A well-elaborated research question may not necessarily be a good question. The proposed study also requires being achievable from both ethical and realistic perspectives, interesting and useful to the clinical practice, and capable to formulate new hypotheses, that may contribute to the generation of knowledge. Researchers have developed an effective way to convey the message of how to build a good research question, that is usually recalled under the acronym of FINER (feasible, interesting, novel, ethical and relevant).[ 5 , 6 , 7 ] Table 2 highlights the main characteristics of FINER criteria.[ 7 ]
Main features of FINER criteria (Feasibility, interest, novelty, ethics, and relevance) to formulate a good research question. Adapted from Cummings et al .[ 7 ]
| Component | Criteria |
|---|---|
| Feasible | -Ensures adequacy of research design -Guarantees adequate funding -Recruits target population strategically -Aims an achievable sample size -Prioritises measurable outcomes -Optimises human and technical resources -Accounts for clinicians commitment -Procures high adherence to the treatment and low rate of dropouts -Opts for appropriate and affordable frame time |
| Interesting | -Engages the interest of principal investigators -Attracts the attention of readers -Presents a different perspective of the problem |
| Novel | -Provides different findings -Generates new hypotheses -Improves methodological flaws of existing studies -Resolves a gap in the existing literature |
| Ethical | -Complies with local ethical committees -Safeguards the main principles of ethical research -Guarantees safety and reversibility of side effects |
| Relevant | -Generates new knowledge -Contributes to improve clinical practice -Stimulates further research -Provides an accurate answer to a specific research question |
Novelty and relevance
Although it is clear that any research project should commence with an accurate literature interpretation, in many instances it represents the start and the end of the research: the reader will soon realise that the answer to several questions can be easily found in the published literature.[ 5 ] When the question overcomes the test of a thorough literature review, the project may become novel (there is a gap in the knowledge, and therefore, there is a need for new evidence on the topic) and relevant (the paper may contribute to change the clinical practice). In this context, it is important to distinguish the difference between statistical significance and clinical relevance: in the aforementioned study of Oba et al .,[ 10 ] despite the means of insertion times were reported as significant for the Supreme™ LMA, as compared with ProSeal™ LMA, the difference found in the insertion times (528 vs. 486 sec, respectively), although reported as significant, had little or no clinical relevance.[ 10 ] Conversely, a statistically significant difference of 12 sec might be of clinical relevance in neonates weighing <5 kg.[ 17 ] Thus, statistical tests must be interpreted in the context of a clinically meaningful effect size, which should be previously defined by the researcher.
Feasibility and ethical aspects
Among FINER criteria, there are two potential barriers that may prevent the successful conduct of the project and publication of the manuscript: feasibility and ethical aspects. These obstacles are usually related to the target population, as discussed above. Feasibility refers not only to the budget but also to the complexity of the design, recruitment strategy, blinding, adequacy of the sample size, measurement of the outcome, time of follow-up of participants, and commitment of clinicians, among others.[ 3 , 7 ] Funding, as a component of feasibility, may also be implicated in the ethical principles of clinical research, because the choice of the primary study question may be markedly influenced by the specific criteria demanded in the interest of potential funders.
Discussing ethical issues with local committees is compulsory, as rules applied might vary among countries.[ 18 ] Potential risks and benefits need to be carefully weighed, based upon the four principles of respect for autonomy, beneficence, non-maleficence, and justice.[ 19 ] Although many of these issues may be related to the population target (e.g., conducting a clinical trial in patients with ongoing cardiopulmonary resuscitation would be inappropriate, as would be anaesthetising patients undergoing elective LASER treatment for condylomas, to examine the performance of supraglottic airway devices),[ 14 , 15 ] ethical conflicts may also arise from the intervention (particularly those involving the occurrence of side effects or complications, and their potential for reversibility), comparison (e.g., use of placebo or sham procedures),[ 19 ] outcome (surrogate outcomes should be considered in lieu of long term outcomes), or time frame (e.g., unnecessary longer exposition to an intervention). Thus, FINER criteria should not be conceived without a concomitant examination of the PICOT checklist, and consequently, PICOT framework and FINER criteria should not be seen as separated components, but rather complementary ingredients of a good research question.
Undoubtedly, no research project can be conducted if it is deemed unfeasible, and most institutional review boards would not be in a position to approve a work with major ethical problems. Nonetheless, whether or not the findings are interesting, is a subjective matter. Engaging the attention of readers also depends upon a number of factors, including the manner of presenting the problem, the background of the topic, the intended audience, and the reader's expectations. Furthermore, the interest is usually linked to the novelty and relevance of the topic, and it is worth nothing that editors and peer reviewers of high-impact medical journals are usually reluctant to accept any publication, if there is no novelty inherent to the research hypothesis, or there is a lack of relevance in the results.[ 11 ] Nevertheless, a considerable number of papers have been published without any novelty or relevance in the topic addressed. This is probably reflected in a recent survey, according to which only a third of respondents declared to have read thoroughly the most recent papers downloaded, and at least half of those manuscripts remained unread.[ 20 ] The same study reported that up to one-third of papers examined remained uncited after 5 years of publication, and only 20% of papers accounted for 80% of the citations.[ 20 ]
Formulating a good research question can be fascinating, albeit challenging, even for experienced investigators. While it is clear that clinical experience in combination with the accurate interpretation of literature and teamwork are essential to develop new ideas, the formulation of a clinical problem usually requires the compliance with PICOT framework in conjunction with FINER criteria, in order to translate a clinical dilemma into a researchable question. Working in the right environment with the adequate support of experienced researchers, will certainly make a difference in the generation of knowledge. By doing this, a lot of time will be saved in the search of the primary study question, and undoubtedly, there will be more chances to become a successful researcher.
Financial support and sponsorship
Conflicts of interest.
There are no conflicts of interest.
Formulating Your Research Question
- First Online: 26 May 2018
Cite this chapter

- Eva O. L. Lantsoght 2 , 3
Part of the book series: Springer Texts in Education ((SPTE))
193k Accesses
In this chapter, the research question is studied. We focus on how to find a research question that is specific enough, so that you are not tempted to explore paths that are only tangentially related to your research question. The literature review identifies gaps in the current knowledge, and you will learn how to frame a research question within these gaps. We then explore how to subdivide the research question into subquestions. These subquestions become the chapters of your dissertation. We also look at creative thinking, a skill necessary to think out of the box to formulate your research question. This chapter discusses how to convince your supervisor of your research question. It can happen that your supervisor already has an idea of the direction in which your research should be going, but if you can provide technically sound arguments based on your literature review why this approach is not ideal, and why you propose a different road, you should be able to have the freedom to explore your proposed option. Once you have outlined your research question, it is necessary to turn the question and subquestions into practical actions. These practical actions link back to the planning skills you learned in Chap. 3 .
This is a preview of subscription content, log in via an institution to check access.
Access this chapter
- Get 10 units per month
- Download Article/Chapter or Ebook
- 1 Unit = 1 Article or 1 Chapter
- Cancel anytime
- Available as PDF
- Read on any device
- Instant download
- Own it forever
- Available as EPUB and PDF
- Compact, lightweight edition
- Dispatched in 3 to 5 business days
- Free shipping worldwide - see info
Tax calculation will be finalised at checkout
Purchases are for personal use only
Institutional subscriptions
Time blocks of 25 minutes during which you concentrate on one single task. You can find more information about the Pomodoro technique in the glossary of Part II.
I often work with noise-cancelling headphones.
The course is sweet and short, and runs frequently. I highly recommend it!
Refer to Chap. 4 for examples on how I use mindmaps to structure documents, such as a literature review report.
Further Reading and References
Kara, H. (2015). Creative research methods in the Social Sciences: A practical guide . Bristol: Policy Press.
Google Scholar
Kara, H. (2015). How to choose your research question. PhD Talk . http://phdtalk.blogspot.com/2015/07/how-to-choose-your-research-question.html
Lantsoght, E. (2012). The creative process: The importance of questions. PhD Talk . http://phdtalk.blogspot.nl/2012/11/the-creative-process-importance-of.html
Feynman, R. P., Leighton, R., & Hutchings, E. (1997). “Surely you’re joking, Mr. Feynman!”: Adventures of a curious character (1st pbk ed.). New York: W.W. Norton.
Lantsoght, E. (2012). The creative process: The creative habit. PhD Talk . http://phdtalk.blogspot.nl/2012/11/the-creative-process-creative-habit.html
Rose, C. (2016). 15 minute history . http://15minutehistory.org /
Oakley, B. (2014). A mind for numbers: How to excel at Math and Science (even if you flunked algebra) . New York: TarcherPerigree.
Lantsoght E (2011). Book review: Starting research: An introduction to academic research and dissertation writing – Roy Preece. PhD Talk . http://phdtalk.blogspot.nl/2011/07/book-review-starting-research.html .
Preece, R. (2000). Starting research: An introduction to academic research and dissertation writing . London: Continuum.
Lantsoght, E. (2014). An example outline diagram for structuring your dissertation. PhD Talk . http://phdtalk.blogspot.nl/2014/08/an-example-outline-diagram-for.html
Download references
Author information
Authors and affiliations.
Universidad San Francisco de Quito, Quito, Ecuador
Eva O. L. Lantsoght
Delft University of Technology, Delft, The Netherlands
You can also search for this author in PubMed Google Scholar
Rights and permissions
Reprints and permissions
Copyright information
© 2018 Springer International Publishing AG, part of Springer Nature
About this chapter
Lantsoght, E.O.L. (2018). Formulating Your Research Question. In: The A-Z of the PhD Trajectory. Springer Texts in Education. Springer, Cham. https://doi.org/10.1007/978-3-319-77425-1_5
Download citation
DOI : https://doi.org/10.1007/978-3-319-77425-1_5
Published : 26 May 2018
Publisher Name : Springer, Cham
Print ISBN : 978-3-319-77424-4
Online ISBN : 978-3-319-77425-1
eBook Packages : Education Education (R0)
Share this chapter
Anyone you share the following link with will be able to read this content:
Sorry, a shareable link is not currently available for this article.
Provided by the Springer Nature SharedIt content-sharing initiative
- Publish with us
Policies and ethics
- Find a journal
- Track your research

IMAGES
VIDEO
COMMENTS
In any quest for knowledge and understanding, asking questions is a fundamental step. When conducting research, the process begins with formulating a research question. A research question acts as a guide for investigation and forms the basis for an essay or a research paper. It defines the scope of the inquiry and helps focus the research efforts.
Formulation of studies question (RQ) is an essentiality earlier than beginning any studies. It targets to discover an present uncertainty in a place of problem and factors to a want for planned investigation. The required details for Research questions in given paragraph.
A good research question is essential to guide your research paper, dissertation, or thesis. All research questions should be: Focused on a single problem or issue. Researchable using primary and/or secondary sources. Feasible to answer within the timeframe and practical constraints. Specific enough to answer thoroughly.
1. Provide a scientific context for the research and to validate it against the 3 criteria for a good research question. 2. Avoid duplication of effort. 3. Identify potential problems in conducting the research. 4 Types of Information for Literature Review. 1. Relevant theories.
In a research paper, the emphasis is on generating a unique question and then synthesizing diverse sources into a coherent essay that supports your argument about the topic. In other words, you integrate information from publications with your own thoughts in order to formulate an argument. Your topic is your starting place: from here, you will ...
Abstract. Formulation of research question (RQ) is an essentiality before starting any research. It aims to explore an existing uncertainty in an area of concern and points to a need for deliberate investigation. It is, therefore, pertinent to formulate a good RQ. The present paper aims to discuss the process of formulation of RQ with stepwise ...
As such, the purpose of this commentary is to provide useful guidance on composing and evaluating rigorous research questions. 2. A framework for formulating research questions. Although every research project is unique, they share common domains that a researcher should consider and define a priori.
Formulating a Research Question As noted in Module 1: Types of Reviews, conducting a "pre-search" is a crucial first step in devising the research question.A well-formulated research question informs the research process. It can focus your information needs (i.e. identify inclusion and exclusion criteria), help to identify key search concepts, and guide you in the direction of relevant ...
contains types of research questions, characteristics, purpose, and develop research questions. Learn with flashcards, games, and more — for free.
The Purpose of Research Questions. Research questions are very important. Both professional researchers and successful student researchers develop research questions. That's because research questions are more than handy tools; they are essential to the research process. By defining exactly what the researcher is trying to find out, these ...
In a research paper, the emphasis is on generating a unique question and then synthesizing diverse sources into a coherent essay that supports your argument about the topic. In other words, you integrate information from publications with your own thoughts in order to formulate an argument. Your topic is your starting place: from here, you will ...
The process of formulating a good research question can be challenging and frustrating. While a comprehensive literature review is compulsory, the researcher usually encounters methodological difficulties in the conduct of the study, particularly if the primary study question has not been adequately selected in accordance with the clinical dilemma that needs to be addressed.
In essence, the research question that guides the sciences and social sciences should do the following three things:2. 1) Post a problem. 2) Shape the problem into a testable hypothesis. 3) Report the results of the tested hypothesis. There are two types of data that can help shape research questions in the sciences and social sciences ...
2.1 Identifying Gaps in the Knowledge. Before you define your research question, you need to have a broad and deep understanding of your research field. In Chap. 4, you learned how to develop the literature review as your starting point for understanding, and then engaging with, the scientific output in your field.
Formulating the right question is not only the first step in doing research but the most important part. In order to succeed, a research question must be the right question having the following characteristics: a question that is interesting a researcher as well the reader; a question that can be answered within the time available; a question that can be translated into a hypothesis; and a ...
The statement that developing research questions and formulating hypotheses are foundational steps that precede the definition of the primary research purpose and specific objectives of a study is true. Research begins with a general question that leads to the modeling of a part of reality in terms of theoretical constructs.
Answer. Answer: Formulation of research question (RQ) is an essentiality before starting any research. ... The researcher then defines what still needs to be known in that particular subject and assesses the implied questions. After narrowing the focus and scope of the research subject, researcher frames a RQ and then evaluates it.
Final answer: Developing a research question is essential for guiding research and focusing inquiry. Explanation: The purpose of developing an overall research question is crucial as it centers and guides the direction of a research plan. By formulating a clear research question, writers can define the scope of their project and provide a focused inquiry for the audience.
What is the reason why formulating a research question is one of the first steps in a research project? See answers Advertisement Advertisement mmanjeetkaurin mmanjeetkaurin Answer: It helps refine what is being researched. It helps lay the groundwork for the research. ... Get the Brainly App Download iOS App
Formulation of research question (RQ) is an essentiality before starting any research. It aims to explore an existing uncertainty in an area of concern and points to a need for deliberate investigation. It is, therefore, pertinent to formulate a good RQ. The present paper aims to discuss the process of formulation of RQ with stepwise approach.
It helps to clarify the scope and relevance of the research and aids in determining its direction and methodology. Yes, in order to formulate the question and purpose for your research requires considering the time dimension. The time dimension refers to the time in which your research is carried out. It has a significant effect on the research ...