An official website of the United States government
The .gov means it’s official. Federal government websites often end in .gov or .mil. Before sharing sensitive information, make sure you’re on a federal government site.
The site is secure. The https:// ensures that you are connecting to the official website and that any information you provide is encrypted and transmitted securely.
- Publications
- Account settings
Preview improvements coming to the PMC website in October 2024. Learn More or Try it out now .
- Advanced Search
- Journal List
- Heart Views
- v.18(3); Jul-Sep 2017

Guidelines To Writing A Clinical Case Report
What is a clinical case report.
A case report is a detailed report of the symptoms, signs, diagnosis, treatment, and follow-up of an individual patient. Case reports usually describe an unusual or novel occurrence and as such, remain one of the cornerstones of medical progress and provide many new ideas in medicine. Some reports contain an extensive review of the relevant literature on the topic. The case report is a rapid short communication between busy clinicians who may not have time or resources to conduct large scale research.
WHAT ARE THE REASONS FOR PUBLISHING A CASE REPORT?
The most common reasons for publishing a case are the following: 1) an unexpected association between diseases or symptoms; 2) an unexpected event in the course observing or treating a patient; 3) findings that shed new light on the possible pathogenesis of a disease or an adverse effect; 4) unique or rare features of a disease; 5) unique therapeutic approaches; variation of anatomical structures.
Most journals publish case reports that deal with one or more of the following:
- Unusual observations
- Adverse response to therapies
- Unusual combination of conditions leading to confusion
- Illustration of a new theory
- Question regarding a current theory
- Personal impact.
STRUCTURE OF A CASE REPORT[ 1 , 2 ]
Different journals have slightly different formats for case reports. It is always a good idea to read some of the target jiurnals case reports to get a general idea of the sequence and format.
In general, all case reports include the following components: an abstract, an introduction, a case, and a discussion. Some journals might require literature review.
The abstract should summarize the case, the problem it addresses, and the message it conveys. Abstracts of case studies are usually very short, preferably not more than 150 words.
Introduction
The introduction gives a brief overview of the problem that the case addresses, citing relevant literature where necessary. The introduction generally ends with a single sentence describing the patient and the basic condition that he or she is suffering from.
This section provides the details of the case in the following order:
- Patient description
- Case history
- Physical examination results
- Results of pathological tests and other investigations
- Treatment plan
- Expected outcome of the treatment plan
- Actual outcome.
The author should ensure that all the relevant details are included and unnecessary ones excluded.
This is the most important part of the case report; the part that will convince the journal that the case is publication worthy. This section should start by expanding on what has been said in the introduction, focusing on why the case is noteworthy and the problem that it addresses.
This is followed by a summary of the existing literature on the topic. (If the journal specifies a separate section on literature review, it should be added before the Discussion). This part describes the existing theories and research findings on the key issue in the patient's condition. The review should narrow down to the source of confusion or the main challenge in the case.
Finally, the case report should be connected to the existing literature, mentioning the message that the case conveys. The author should explain whether this corroborates with or detracts from current beliefs about the problem and how this evidence can add value to future clinical practice.
A case report ends with a conclusion or with summary points, depending on the journal's specified format. This section should briefly give readers the key points covered in the case report. Here, the author can give suggestions and recommendations to clinicians, teachers, or researchers. Some journals do not want a separate section for the conclusion: it can then be the concluding paragraph of the Discussion section.
Notes on patient consent
Informed consent in an ethical requirement for most studies involving humans, so before you start writing your case report, take a written consent from the patient as all journals require that you provide it at the time of manuscript submission. In case the patient is a minor, parental consent is required. For adults who are unable to consent to investigation or treatment, consent of closest family members is required.
Patient anonymity is also an important requirement. Remember not to disclose any information that might reveal the identity of the patient. You need to be particularly careful with pictures, and ensure that pictures of the affected area do not reveal the identity of the patient.

Writing a Case Report
This page is intended for medical students, residents or others who do not have much experience with case reports, but are planning on writing one.
What is a case report? A medical case report, also known as a case study, is a detailed description of a clinical encounter with a patient. The most important aspect of a case report, i.e. the reason you would go to the trouble of writing one, is that the case is sufficiently unique, rare or interesting such that other medical professionals will learn something from it.
Case reports are commonly of the following categories :
- Rare diseases
- Unusual presentation of disease
- Unexpected events
- Unusual combination of diseases or conditions
- Difficult or inconclusive diagnosis
- Treatment or management challenges
- Personal impact
- Observations that shed new light on a disease or condition
- Anatomical variations
It is important that you recognize what is unique or interesting about your case, and this must be described clearly in the case report.
Case reports generally take the format of :
1. Background
2. Case presentation
3. Observations and investigation
4. Diagnosis
5. Treatment
7. Discussion
Does a case report require IRB approval?
Case reports typically discuss a single patient. If this is true for your case report, then it most likely does not require IRB approval because it not considered research. If you have more than one patient, your study could qualify as a Case Series, which would require IRB review. If you have questions, you chould check your local IRB's guidelines on reviewing case reports.
Are there other rules for writing a case report?
First, you will be collecting protected health information, thus HIPAA applies to case reports. Spectrum Health has created a very helpful guidance document for case reports, which you can see here: Case Report Guidance - Spectrum Health
While this guidance document was created by Spectrum Health, the rules and regulations outlined could apply to any case report. This includes answering questions like: Do I need written HIPAA authorization to publish a case report? When do I need IRB review of a case report? What qualifies as a patient identifier?
How do I get started?
1. We STRONGLY encourage you to consult the CARE Guidelines, which provide guidance on writing case reports - https://www.care-statement.org/
Specifically, the checklist - https://www.care-statement.org/checklist - which explains exactly the information you should collect and include in your case report.
2. Identify a case. If you are a medical student, you may not yet have the clinical expertise to determine if a specific case is worth writing up. If so, you must seek the help of a clinician. It is common for students to ask attendings or residents if they have any interesting cases that can be used for a case report.
3. Select a journal or two to which you think you will submit the case report. Journals often have specific requirements for publishing case reports, which could include a requirement for informed consent, a letter or statement from the IRB and other things. Journals may also charge publication fees (see Is it free to publish? below)
4. Obtain informed consent from the patient (see " Do I have to obtain informed consent from the patient? " below). Journals may have their own informed consent form that they would like you to use, so please look for this when selecting a journal.
Once you've identified the case, selected an appropriate journal(s), and considered informed consent, you can collect the required information to write the case report.
How do I write a case report?
Once you identify a case and have learned what information to include in the case report, try to find a previously published case report. Finding published case reports in a similar field will provide examples to guide you through the process of writing a case report.
One journal you can consult is BMJ Case Reports . MSU has an institutional fellowship with BMJ Case Reports which allows MSU faculty, staff and students to publish in this journal for free. See this page for a link to the journal and more information on publishing- https://lib.msu.edu/medicalwriting_publishing/
There are numerous other journals where you can find published case reports to help guide you in your writing.
Do I have to obtain informed consent from the patient?
The CARE guidelines recommend obtaining informed consent from patients for all case reports. Our recommendation is to obtain informed consent from the patient. Although not technically required, especially if the case report does not include any identifying information, some journals require informed consent for all case reports before publishing. The CARE guidelines recommend obtaining informed consent AND the patient's perspective on the treatment/outcome (if possible). Please consider this as well.
If required, it is recommended you obtain informed consent before the case report is written.
An example of a case report consent form can be found on the BMJ Case Reports website, which you can access via the MSU library page - https://casereports.bmj.com/ . Go to "Instructions for Authors" and then "Patient Consent" to find the consent form they use. You can create a similar form to obtain consent from your patient. If you have identified a journal already, please consult their requirements and determine if they have a specific consent form they would like you to use.
Seek feedback
Once you have written a draft of the case report, you should seek feedback on your writing, from experts in the field if possible, or from those who have written case reports before.
Selecting a journal
Aside from BMJ Case Reports mentioned above, there are many, many journals out there who publish medical case reports. Ask your mentor if they have a journal they would like to use. If you need to select on your own, here are some strategies:
1. Do a PubMed search. https://pubmed.ncbi.nlm.nih.gov/
a. Do a search for a topic, disease or other feature of your case report
b. When the results appear, on the left side of the page is a limiter for "article type". Case reports are an article type to which you can limit your search results. If you don't see that option on the left, click "additional filters".
c. Review the case reports that come up and see what journals they are published in.
2. Use JANE - https://jane.biosemantics.org/
3. Check with specialty societies. Many specialty societies are affiliated with one or more journal, which can be reviewed for ones that match your needs
4. Search through individual publisher journal lists. Elsevier publishes many different medical research journals, and they have a journal finder, much like JANE ( https://journalfinder.elsevier.com/ ). This is exclusive to Elsevier journals. There are many other publishers of medical journals for review, including Springer, Dove Press, BMJ, BMC, Wiley, Sage, Nature and many others.
Is it free to publish ?
Be aware that it may not be free to publish your case report. Many journals charge publication fees. Of note, many open access journals charge author fees of thousands of dollars. Other journals have smaller page charges (i.e. $60 per page), and still others will publish for free, with an "open access option". It is best practice to check the journal's Info for Authors section or Author Center to determine what the cost is to publish. MSU-CHM does NOT have funds to support publication costs, so this is an important step if you do not want to pay out of pocket for publishing
*A more thorough discussion on finding a journal, publication costs, predatory journals and other publication-related issues can be found here: https://research.chm.msu.edu/students-residents/finding-a-journal
Gagnier JJ, Kienle G, Altman DG, Moher D, Sox H, Riley D. 2013. The CARE guidelines: Consensus-based clinical case reporting guideline development. Glob Adv Health Med . 2:38-43. doi: 10.7453/gahmj.2013.008
Riley DS, Barber MS, Kienle GS, AronsonJK, von Schoen-Angerer T, Tugwell P, Kiene H, Helfand M, Altman DG, Sox H, Werthmann PG, Moher D, Rison RA, Shamseer L, Koch CA, Sun GH, Hanaway P, Sudak NL, Kaszkin-Bettag M, Carpenter JE, Gagnier JJ. 2017. CARE guidelines for case reports: explanation and elaboration document . J Clin Epidemiol . 89:218-234. doi: 10.1016/j.jclinepi.2017.04.026
Guidelines to writing a clinical case report. 2017. Heart Views . 18:104-105. doi: 10.4103/1995-705X.217857
Ortega-Loubon C, Culquichicon C, Correa R. The importance of writing and publishing case reports during medical education. 2017. Cureus. 9:e1964. doi: 10.7759/cureus.1964
Writing and publishing a useful and interesting case report. 2019. BMJ Case Reports. https://casereports.bmj.com/pages/wp-content/uploads/sites/69/2019/04/How-to-write-a-Case-Report-DIGITAL.pdf
Camm CF. Writing an excellent case report: EHJ Case Reports , Case of the Year 2019. 2020. European Heart Jounrnal. 41:1230-1231. https://doi.org/10.1093/eurheartj/ehaa176
*content developed by Mark Trottier, PhD
Thank you for visiting nature.com. You are using a browser version with limited support for CSS. To obtain the best experience, we recommend you use a more up to date browser (or turn off compatibility mode in Internet Explorer). In the meantime, to ensure continued support, we are displaying the site without styles and JavaScript.
- View all journals
- Explore content
- About the journal
- Publish with us
- Sign up for alerts
- Published: 30 January 2023
A student guide to writing a case report
- Maeve McAllister 1
BDJ Student volume 30 , pages 12–13 ( 2023 ) Cite this article
25 Accesses
Metrics details
As a student, it can be hard to know where to start when reading or writing a clinical case report either for university or out of special interest in a Journal. I have collated five top tips for writing an insightful and relevant case report.
A case report is a structured report of the clinical process of a patient's diagnostic pathway, including symptoms, signs, diagnosis, treatment planning (short and long term), clinical outcomes and follow-up. 1 Some of these case reports can sometimes have simple titles, to the more unusual, for example, 'Oral Tuberculosis', 'The escapee wisdom tooth', 'A difficult diagnosis'. They normally begin with the word 'Sir' and follow an introduction from this.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
We are sorry, but there is no personal subscription option available for your country.
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Guidelines To Writing a Clinical Case Report. Heart Views 2017; 18 , 104-105.
British Dental Journal. Case reports. Available online at: www.nature.com/bdj/articles?searchType=journalSearch&sort=PubDate&type=case-report&page=2 (accessed August 17, 2022).
Chate R, Chate C. Achenbach's syndrome. Br Dent J 2021; 231: 147.
Abdulgani A, Muhamad, A-H and Watted N. Dental case report for publication; step by step. J Dent Med Sci 2014; 3 : 94-100.
Download references
Author information
Authors and affiliations.
Queen´s University Belfast, Belfast, United Kingdom
Maeve McAllister
You can also search for this author in PubMed Google Scholar
Corresponding author
Correspondence to Maeve McAllister .
Rights and permissions
Reprints and permissions
About this article
Cite this article.
McAllister, M. A student guide to writing a case report. BDJ Student 30 , 12–13 (2023). https://doi.org/10.1038/s41406-023-0925-y
Download citation
Published : 30 January 2023
Issue Date : 30 January 2023
DOI : https://doi.org/10.1038/s41406-023-0925-y
Share this article
Anyone you share the following link with will be able to read this content:
Sorry, a shareable link is not currently available for this article.
Provided by the Springer Nature SharedIt content-sharing initiative
Quick links
- Explore articles by subject
- Guide to authors
- Editorial policies
- PRO Courses Guides New Tech Help Pro Expert Videos About wikiHow Pro Upgrade Sign In
- EDIT Edit this Article
- EXPLORE Tech Help Pro About Us Random Article Quizzes Request a New Article Community Dashboard This Or That Game Popular Categories Arts and Entertainment Artwork Books Movies Computers and Electronics Computers Phone Skills Technology Hacks Health Men's Health Mental Health Women's Health Relationships Dating Love Relationship Issues Hobbies and Crafts Crafts Drawing Games Education & Communication Communication Skills Personal Development Studying Personal Care and Style Fashion Hair Care Personal Hygiene Youth Personal Care School Stuff Dating All Categories Arts and Entertainment Finance and Business Home and Garden Relationship Quizzes Cars & Other Vehicles Food and Entertaining Personal Care and Style Sports and Fitness Computers and Electronics Health Pets and Animals Travel Education & Communication Hobbies and Crafts Philosophy and Religion Work World Family Life Holidays and Traditions Relationships Youth
- Browse Articles
- Learn Something New
- Quizzes Hot
- This Or That Game
- Train Your Brain
- Explore More
- Support wikiHow
- About wikiHow
- Log in / Sign up
- Education and Communications
- Medical Studies
How to Write a Medical Case Study Report
Last Updated: April 18, 2024 Fact Checked
This article was medically reviewed by Mark Ziats, MD, PhD and by wikiHow staff writer, Jennifer Mueller, JD . Dr. Mark Ziats is an Internal Medicine Physician, Scientist, Entrepreneur, and the Medical Director of xBiotech. With over five years of experience, he specializes in biotechnology, genomics, and medical devices. He earned a Doctor of Medicine degree from Baylor College of Medicine, a Ph.D. in Genetics from the University of Cambridge, and a BS in Biochemistry and Chemistry from Clemson University. He also completed the INNoVATE Program in Biotechnology Entrepreneurship at The Johns Hopkins University - Carey Business School. Dr. Ziats is board certified by the American Board of Internal Medicine. There are 15 references cited in this article, which can be found at the bottom of the page. This article has been fact-checked, ensuring the accuracy of any cited facts and confirming the authority of its sources. This article has been viewed 187,337 times.
You've encountered an interesting and unusual case on your rounds, and a colleague or supervising physician says, "Why don't you write up a case study report?" If you've never written one before, that might sound intimidating, but it's a great way to get started in medical writing. Case studies always follow a standard structure and format, so the writing is very formulaic once you get the hang of it. Read on for a step-by-step guide to writing your first case study report.
What is a case study report?

- Medical students or residents typically do the bulk of the writing of the report. If you're just starting your medical career, a case study report is a great way to get a publication under your belt. [2] X Research source

- If the patient is a minor or is incapable of giving informed consent, get consent from their parents or closest relative. [4] X Trustworthy Source PubMed Central Journal archive from the U.S. National Institutes of Health Go to source
- Your hospital likely has specific consent forms to use. Ask your supervising physician if you're not sure where to get one.
- Some journals also have their own consent form. Check your target journal's author or submission information to make sure. [5] X Research source
How is a case study report structured?

- Even though the introduction is the first part of a case study report, doctors typically write it last. You'll have a better idea of how to introduce your case study to readers after you've written it.
- Your abstract comes at the top, before the introduction, and provides a brief summary of the entire report. Unless your case study is published in an open-access journal, the abstract is the only part of the article many readers will see.

- Many journals offer templates and checklists you can use to make sure your case study includes everything necessary and is formatted properly—take advantage of these! Some journals, such as BMJ Case Reports , require all case studies submitted to use their templates.
Drafting Your Medical Case Study Report

- Patient description
- Chronological case history
- Physical exam results
- Results of any pathological tests, imaging, or other investigations
- Treatment plan
- Expected outcome of treatment
- Actual outcome of treatment

- Why the patient sought medical help (you can even use their own words)
- Important information that helped you settle on your diagnosis
- The results of your clinical examination, including diagnostic tests and their results, along with any helpful images
- A description of the treatment plan
- The outcome, including how and why treatment ended and how long the patient was under your care [11] X Trustworthy Source PubMed Central Journal archive from the U.S. National Institutes of Health Go to source

- You will need references to back up symptoms of the condition, common treatment, and the expected outcome of that common treatment.
- Use your research to paint a picture of the usual case of a patient with a similar condition—it'll help you show how unusual and different your patient's case is.
- Generally, aim for around 20 references—no fewer than 15, but no more than 25. [13] X Trustworthy Source PubMed Central Journal archive from the U.S. National Institutes of Health Go to source

- Close your discussion section with a summary of the lessons learned from the case and why it's significant to consider when treating similar cases in the future.
- Outline any open questions that remain. You might also provide suggestions for future research.

- In your conclusion, you might also give suggestions or recommendations to readers based on what you learned as a result of the case.
- Some journals don't want a separate conclusion section. If that's the case for one of your target journals, just move this paragraph to the end of your discussion section.
Polishing Your Report for Submission to Publishers

- Most titles are fewer than 10 words long and include the name of the disease or condition treated.
- You might also include the treatment used and whether the outcome was successful. When deciding what to include, think about the reason you wrote the case study in the first place and why you think it's important for other clinicians to read.

- Made a significant intellectual contribution to the case study report
- Was involved in the medical care of the patient reported
- Can explain and defend the data presented in the report
- Has approved the final manuscript before submission for publication

- Keep in mind that the abstract is not just going to be the first thing people read—it will often be the only thing people read. Make sure that if someone is going to walk away having only read the abstract, they'll still get the same message they would have if they read the whole thing.
- There are 2 basic types of abstract: narrative and structured. A narrative abstract is a single paragraph written in narrative prose. A structured abstract includes headings that correspond with the sections of the paper, then a brief summary of each section. Use the format preferred by your target journal.

- Look for keywords that are relevant to your field or sub-field and directly related to the content of your article, such as the name of the condition or specific treatments you used.
- Most journals allow 4-8 keywords but check the submission guidelines of your target journal to make sure.

- Blur out the patient's face as well as any tattoos, birthmarks, or unrelated scars that are visible in diagnostic images.

- It's common to thank the patient, but that's up to you. Even if you don't, include a statement indicating that you have the patient's written, informed consent to publish the information.
- Read the journal's submission guidelines for a definition of what that journal considers a conflict of interest. They're generally the same, but some might be stricter than others. [22] X Research source

- If you're not familiar with the citation style used by your target journal, check online for a guide. There might also be one available at your hospital or medical school library.
- Medical librarians can also help with citation style and references if you run into something tricky—don't just wing it! Correct citation style insures that readers can access the materials you cite.

- It's also a good idea to get a beta reader who isn't a medical professional. Their comments can help you figure out where you need to clarify your points.
- Read a lot of case studies published in your target journals—it will help you internalize the tone and style that journal is looking for.
Submitting Your Report to Publishers

- Look into the background and reputation of journals before you decide to submit to them. Only seek publication from reputable journals in which articles go through a peer-review process.
- Find out what publishing fees the journals charge. Keep in mind that open-access journals tend to charge higher publishing fees. [26] X Research source
- Read each journal's submission and editorial guidelines carefully. They'll tell you exactly how to format your case study, how long each section should be, and what citation style to use. [27] X Research source
- For electronic journals that only publish case reports, try BMJ Case Reports , Journal of Medical Case Reports , or Radiology Case Reports .

- If your manuscript isn't suitable for the journal you submitted to, the journal might offer to forward it to an associated journal where it would be a better fit.
- When your manuscript is provisionally accepted, the journal will send it to other doctors for evaluation under the peer-review process.
- Most medical journals don't accept simultaneous submissions, meaning you'll have to submit to your first choice, wait for their decision, then move to the next journal on the list if they don't bite.

- Along with your revised manuscript, include a letter with your response to each of the reviewer's comments. Where you made revisions, add page numbers to indicate where the revisions are that address that reviewer's comments.
- Sometimes, doctors involved in the peer review process will indicate that the journal should reject the manuscript. If that's the case, you'll get a letter explaining why your case study report won't be published and you're free to submit it elsewhere.

- Some journals require you to have your article professionally copy-edited at your own cost while others do this in-house. The editors will let you know what you're responsible for.

- With your acceptance letter, you'll get instructions on how to make payment and how much you owe. Take note of the deadline and make sure you pay it as soon as possible to avoid publication delays.
- Some journals will publish for free, with an "open-access option" that allows you to pay a fee only if you want open access to your article. [32] X Research source

- Through the publishing agreement, you assign your copyright in the article to the journal. This allows the journal to legally publish your work. That assignment can be exclusive or non-exclusive and may only last for a specific term. Read these details carefully!
- If you published an open-access article, you don't assign the copyright to the publisher. The publishing agreement merely gives the journal the right to publish the "Version of Record." [33] X Research source
How do I find a suitable case for a report?

- A rare disease, or unusual presentation of any disease
- An unusual combination of diseases or conditions
- A difficult or inconclusive diagnosis
- Unexpected developments or responses to treatment
- Personal impact
- Observations that shed new light on the patient's disease or condition

- There might be other members of your medical team that want to help with writing. If so, use one of these brainstorming sessions to divvy up writing responsibilities in a way that makes the most sense given your relative skills and experience.
- Senior doctors might also be able to name some journals that would potentially publish your case study. [36] X Research source
Expert Q&A
You Might Also Like

- ↑ https://www.elsevier.com/connect/authors-update/the-dos-and-donts-of-writing-and-publishing-case-reports
- ↑ https://www.bmj.com/content/350/bmj.h2693
- ↑ https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5686928/
- ↑ https://health.usf.edu/medicine/internalmedicine/im-impact/~/media/B3A3421F4C144FA090AE965C21791A3C.ashx
- ↑ https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2597880/
- ↑ https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6476221/
- ↑ https://www.springer.com/gp/authors-editors/authorandreviewertutorials/writing-a-journal-manuscript/title-abstract-and-keywords/10285522
- ↑ http://www.ncbi.nlm.nih.gov/pmc/articles/PMC2597880/
- ↑ https://thelancet.com/pb/assets/raw/Lancet/authors/tl-info-for-authors.pdf
- ↑ https://jmedicalcasereports.biomedcentral.com/articles/10.1186/s13256-017-1351-y
- ↑ https://guides.himmelfarb.gwu.edu/casereports
- ↑ https://casereports.bmj.com/pages/authors/
- ↑ https://jmedicalcasereports.biomedcentral.com/articles/10.1186/1752-1947-7-239
- ↑ https://research.chm.msu.edu/students-residents/writing-a-case-report
- ↑ https://authorservices.taylorandfrancis.com/publishing-your-research/moving-through-production/copyright-for-journal-authors/#
About This Article

Medical Disclaimer
The content of this article is not intended to be a substitute for professional medical advice, examination, diagnosis, or treatment. You should always contact your doctor or other qualified healthcare professional before starting, changing, or stopping any kind of health treatment.
Read More...
To start a medical case study report, first choose a title that clearly reflects the contents of the report. You’ll also need to list any participating authors and develop a list of keywords, as well as an abstract summarizing the report. Your report will need to include an introduction summarizing the context of the report, as well as a detailed presentation of the case. Don’t forget to include a thorough citation list and acknowledgements of anyone else who participated in the study. For more tips from our Medical co-author, including how to get your case study report published, keep reading! Did this summary help you? Yes No
- Send fan mail to authors
Reader Success Stories
Sep 5, 2020
Did this article help you?
Asfia Banu Pasha
Apr 10, 2017
Jun 20, 2021
Mar 1, 2017

Featured Articles

Trending Articles

Watch Articles

- Terms of Use
- Privacy Policy
- Do Not Sell or Share My Info
- Not Selling Info
wikiHow Tech Help Pro:
Level up your tech skills and stay ahead of the curve
- Search Menu
- Sign in through your institution
- Advance articles
- AHFS First Release
- AJHP Voices
- AJHP Residents Edition
- Top Twenty-Five Articles
- ASHP National Surveys of Pharmacy Practice in Hospital Settings
- Medication Safety
- Pharmacy Technicians
- Specialty Pharmacy
- Emergency Preparedness and Clinician Well-being
- Author Guidelines
- Submission Site
- Open Access
- Information for Reviewers
- Self-Archiving Policy
- Author Instructions for Residents Edition
- Advertising and Corporate Services
- Advertising
- Reprints and ePrints
- Sponsored Supplements
- Editorial Board
- Permissions
- Journals on Oxford Academic
- Books on Oxford Academic
- < Previous
How to write a patient case report
- Article contents
- Figures & tables
- Supplementary Data
Henry Cohen, How to write a patient case report, American Journal of Health-System Pharmacy , Volume 63, Issue 19, 1 October 2006, Pages 1888–1892, https://doi.org/10.2146/ajhp060182
- Permissions Icon Permissions
Purpose. Guidelines for writing patient case reports, with a focus on medication-related reports, are provided.
Summary. The format of a patient case report encompasses the following five sections: an abstract, an introduction and objective that contain a literature review, a description of the case report, a discussion that includes a detailed explanation of the literature review, a summary of the case, and a conclusion. The abstract of a patient case report should succinctly include the four sections of the main text of the report. The introduction section should provide the subject, purpose, and merit of the case report. It must explain why the case report is novel or merits review, and it should include a comprehensive literature review that corroborates the author’s claims. The case presentation section should describe the case in chronological order and in enough detail for the reader to establish his or her own conclusions about the case’s validity. The discussion section is the most important section of the case report. It ought to evaluate the patient case for accuracy, validity, and uniqueness; compare and contrast the case report with the published literature; derive new knowledge; summarize the essential features of the report; and draw recommendations. The conclusion section should be brief and provide a conclusion with evidence-based recommendations and applicability to practice.
Conclusion. Patient case reports are valuable resources of new and unusual information that may lead to vital research.
Email alerts
Citing articles via.
- Recommend to Your Librarian
Affiliations
- Online ISSN 1535-2900
- Print ISSN 1079-2082
- Copyright © 2024 American Society of Health-System Pharmacists
- About Oxford Academic
- Publish journals with us
- University press partners
- What we publish
- New features
- Open access
- Institutional account management
- Rights and permissions
- Get help with access
- Accessibility
- Media enquiries
- Oxford University Press
- Oxford Languages
- University of Oxford
Oxford University Press is a department of the University of Oxford. It furthers the University's objective of excellence in research, scholarship, and education by publishing worldwide
- Copyright © 2024 Oxford University Press
- Cookie settings
- Cookie policy
- Privacy policy
- Legal notice
This Feature Is Available To Subscribers Only
Sign In or Create an Account
This PDF is available to Subscribers Only
For full access to this pdf, sign in to an existing account, or purchase an annual subscription.
- - Google Chrome
Intended for healthcare professionals
- My email alerts
- BMA member login
- Username * Password * Forgot your log in details? Need to activate BMA Member Log In Log in via OpenAthens Log in via your institution

Search form
- Advanced search
- Search responses
- Search blogs
- How to present patient...
How to present patient cases
- Related content
- Peer review
- Mary Ni Lochlainn , foundation year 2 doctor 1 ,
- Ibrahim Balogun , healthcare of older people/stroke medicine consultant 1
- 1 East Kent Foundation Trust, UK
A guide on how to structure a case presentation
This article contains...
-History of presenting problem
-Medical and surgical history
-Drugs, including allergies to drugs
-Family history
-Social history
-Review of systems
-Findings on examination, including vital signs and observations
-Differential diagnosis/impression
-Investigations
-Management
Presenting patient cases is a key part of everyday clinical practice. A well delivered presentation has the potential to facilitate patient care and improve efficiency on ward rounds, as well as a means of teaching and assessing clinical competence. 1
The purpose of a case presentation is to communicate your diagnostic reasoning to the listener, so that he or she has a clear picture of the patient’s condition and further management can be planned accordingly. 2 To give a high quality presentation you need to take a thorough history. Consultants make decisions about patient care based on information presented to them by junior members of the team, so the importance of accurately presenting your patient cannot be overemphasised.
As a medical student, you are likely to be asked to present in numerous settings. A formal case presentation may take place at a teaching session or even at a conference or scientific meeting. These presentations are usually thorough and have an accompanying PowerPoint presentation or poster. More often, case presentations take place on the wards or over the phone and tend to be brief, using only memory or short, handwritten notes as an aid.
Everyone has their own presenting style, and the context of the presentation will determine how much detail you need to put in. You should anticipate what information your senior colleagues will need to know about the patient’s history and the care he or she has received since admission, to enable them to make further management decisions. In this article, I use a fictitious case to show how you can structure case presentations, which can be adapted to different clinical and teaching settings (box 1).
Box 1: Structure for presenting patient cases
Presenting problem, history of presenting problem, medical and surgical history.
Drugs, including allergies to drugs
Family history
Social history, review of systems.
Findings on examination, including vital signs and observations
Differential diagnosis/impression
Investigations
Case: tom murphy.
You should start with a sentence that includes the patient’s name, sex (Mr/Ms), age, and presenting symptoms. In your presentation, you may want to include the patient’s main diagnosis if known—for example, “admitted with shortness of breath on a background of COPD [chronic obstructive pulmonary disease].” You should include any additional information that might give the presentation of symptoms further context, such as the patient’s profession, ethnic origin, recent travel, or chronic conditions.
“ Mr Tom Murphy is a 56 year old ex-smoker admitted with sudden onset central crushing chest pain that radiated down his left arm.”
In this section you should expand on the presenting problem. Use the SOCRATES mnemonic to help describe the pain (see box 2). If the patient has multiple problems, describe each in turn, covering one system at a time.
Box 2: SOCRATES—mnemonic for pain
Associations
Time course
Exacerbating/relieving factors
“ The pain started suddenly at 1 pm, when Mr Murphy was at his desk. The pain was dull in nature, and radiated down his left arm. He experienced shortness of breath and felt sweaty and clammy. His colleague phoned an ambulance. He rated the pain 9/10 in severity. In the ambulance he was given GTN [glyceryl trinitrate] spray under the tongue, which relieved the pain to 5/10. The pain lasted 30 minutes in total. No exacerbating factors were noted. Of note: Mr Murphy is an ex-smoker with a 20 pack year history”
Some patients have multiple comorbidities, and the most life threatening conditions should be mentioned first. They can also be categorised by organ system—for example, “has a long history of cardiovascular disease, having had a stroke, two TIAs [transient ischaemic attacks], and previous ACS [acute coronary syndrome].” For some conditions it can be worth stating whether a general practitioner or a specialist manages it, as this gives an indication of its severity.
In a surgical case, colleagues will be interested in exercise tolerance and any comorbidity that could affect the patient’s fitness for surgery and anaesthesia. If the patient has had any previous surgical procedures, mention whether there were any complications or reactions to anaesthesia.
“Mr Murphy has a history of type 2 diabetes, well controlled on metformin. He also has hypertension, managed with ramipril, and gout. Of note: he has no history of ischaemic heart disease (relevant negative) (see box 3).”
Box 3: Relevant negatives
Mention any relevant negatives that will help narrow down the differential diagnosis or could be important in the management of the patient, 3 such as any risk factors you know for the condition and any associations that you are aware of. For example, if the differential diagnosis includes a condition that you know can be hereditary, a relevant negative could be the lack of a family history. If the differential diagnosis includes cardiovascular disease, mention the cardiovascular risk factors such as body mass index, smoking, and high cholesterol.
Highlight any recent changes to the patient’s drugs because these could be a factor in the presenting problem. Mention any allergies to drugs or the patient’s non-compliance to a previously prescribed drug regimen.
To link the medical history and the drugs you might comment on them together, either here or in the medical history. “Mrs Walsh’s drugs include regular azathioprine for her rheumatoid arthritis.”Or, “His regular drugs are ramipril 5 mg once a day, metformin 1g three times a day, and allopurinol 200 mg once a day. He has no known drug allergies.”
If the family history is unrelated to the presenting problem, it is sufficient to say “no relevant family history noted.” For hereditary conditions more detail is needed.
“ Mr Murphy’s father experienced a fatal myocardial infarction aged 50.”
Social history should include the patient’s occupation; their smoking, alcohol, and illicit drug status; who they live with; their relationship status; and their sexual history, baseline mobility, and travel history. In an older patient, more detail is usually required, including whether or not they have carers, how often the carers help, and if they need to use walking aids.
“He works as an accountant and is an ex-smoker since five years ago with a 20 pack year history. He drinks about 14 units of alcohol a week. He denies any illicit drug use. He lives with his wife in a two storey house and is independent in all activities of daily living.”
Do not dwell on this section. If something comes up that is relevant to the presenting problem, it should be mentioned in the history of the presenting problem rather than here.
“Systems review showed long standing occasional lower back pain, responsive to paracetamol.”
Findings on examination
Initially, it can be useful to practise presenting the full examination to make sure you don’t leave anything out, but it is rare that you would need to present all the normal findings. Instead, focus on the most important main findings and any abnormalities.
“On examination the patient was comfortable at rest, heart sounds one and two were heard with no additional murmurs, heaves, or thrills. Jugular venous pressure was not raised. No peripheral oedema was noted and calves were soft and non-tender. Chest was clear on auscultation. Abdomen was soft and non-tender and normal bowel sounds were heard. GCS [Glasgow coma scale] was 15, pupils were equal and reactive to light [PEARL], cranial nerves 1-12 were intact, and he was moving all four limbs. Observations showed an early warning score of 1 for a tachycardia of 105 beats/ min. Blood pressure was 150/90 mm Hg, respiratory rate 18 breaths/min, saturations were 98% on room air, and he was apyrexial with a temperature of 36.8 ºC.”
Differential diagnoses
Mentioning one or two of the most likely diagnoses is sufficient. A useful phrase you can use is, “I would like to rule out,” especially when you suspect a more serious cause is in the differential diagnosis. “History and examination were in keeping with diverticular disease; however, I would like to rule out colorectal cancer in this patient.”
Remember common things are common, so try not to mention rare conditions first. Sometimes it is acceptable to report investigations you would do first, and then base your differential diagnosis on what the history and investigation findings tell you.
“My impression is acute coronary syndrome. The differential diagnosis includes other cardiovascular causes such as acute pericarditis, myocarditis, aortic stenosis, aortic dissection, and pulmonary embolism. Possible respiratory causes include pneumonia or pneumothorax. Gastrointestinal causes include oesophageal spasm, oesophagitis, gastro-oesophageal reflux disease, gastritis, cholecystitis, and acute pancreatitis. I would also consider a musculoskeletal cause for the pain.”
This section can include a summary of the investigations already performed and further investigations that you would like to request. “On the basis of these differentials, I would like to carry out the following investigations: 12 lead electrocardiography and blood tests, including full blood count, urea and electrolytes, clotting screen, troponin levels, lipid profile, and glycated haemoglobin levels. I would also book a chest radiograph and check the patient’s point of care blood glucose level.”
You should consider recommending investigations in a structured way, prioritising them by how long they take to perform and how easy it is to get them done and how long it takes for the results to come back. Put the quickest and easiest first: so bedside tests, electrocardiography, followed by blood tests, plain radiology, then special tests. You should always be able to explain why you would like to request a test. Mention the patient’s baseline test values if they are available, especially if the patient has a chronic condition—for example, give the patient’s creatinine levels if he or she has chronic kidney disease This shows the change over time and indicates the severity of the patient’s current condition.
“To further investigate these differentials, 12 lead electrocardiography was carried out, which showed ST segment depression in the anterior leads. Results of laboratory tests showed an initial troponin level of 85 µg/L, which increased to 1250 µg/L when repeated at six hours. Blood test results showed raised total cholesterol at 7.6 mmol /L and nil else. A chest radiograph showed clear lung fields. Blood glucose level was 6.3 mmol/L; a glycated haemoglobin test result is pending.”
Dependent on the case, you may need to describe the management plan so far or what further management you would recommend.“My management plan for this patient includes ACS [acute coronary syndrome] protocol, echocardiography, cardiology review, and treatment with high dose statins. If you are unsure what the management should be, you should say that you would discuss further with senior colleagues and the patient. At this point, check to see if there is a treatment escalation plan or a “do not attempt to resuscitate” order in place.
“Mr Murphy was given ACS protocol in the emergency department. An echocardiogram has been requested and he has been discussed with cardiology, who are going to come and see him. He has also been started on atorvastatin 80 mg nightly. Mr Murphy and his family are happy with this plan.”
The summary can be a concise recap of what you have presented beforehand or it can sometimes form a standalone presentation. Pick out salient points, such as positive findings—but also draw conclusions from what you highlight. Finish with a brief synopsis of the current situation (“currently pain free”) and next step (“awaiting cardiology review”). Do not trail off at the end, and state the diagnosis if you are confident you know what it is. If you are not sure what the diagnosis is then communicate this uncertainty and do not pretend to be more confident than you are. When possible, you should include the patient’s thoughts about the diagnosis, how they are feeling generally, and if they are happy with the management plan.
“In summary, Mr Murphy is a 56 year old man admitted with central crushing chest pain, radiating down his left arm, of 30 minutes’ duration. His cardiac risk factors include 20 pack year smoking history, positive family history, type 2 diabetes, and hypertension. Examination was normal other than tachycardia. However, 12 lead electrocardiography showed ST segment depression in the anterior leads and troponin rise from 85 to 250 µg/L. Acute coronary syndrome protocol was initiated and a diagnosis of NSTEMI [non-ST elevation myocardial infarction] was made. Mr Murphy is currently pain free and awaiting cardiology review.”
Originally published as: Student BMJ 2017;25:i4406
Competing interests: None declared.
Provenance and peer review: Not commissioned; externally peer reviewed
- ↵ Green EH, Durning SJ, DeCherrie L, Fagan MJ, Sharpe B, Hershman W. Expectations for oral case presentations for clinical clerks: opinions of internal medicine clerkship directors. J Gen Intern Med 2009 ; 24 : 370 - 3 . doi:10.1007/s11606-008-0900-x pmid:19139965 . OpenUrl CrossRef PubMed Web of Science
- ↵ Olaitan A, Okunade O, Corne J. How to present clinical cases. Student BMJ 2010;18:c1539.
- ↵ Gaillard F. The secret art of relevant negatives, Radiopedia 2016; http://radiopaedia.org/blog/the-secret-art-of-relevant-negatives .
Case Study Research Method in Psychology
Saul Mcleod, PhD
Editor-in-Chief for Simply Psychology
BSc (Hons) Psychology, MRes, PhD, University of Manchester
Saul Mcleod, PhD., is a qualified psychology teacher with over 18 years of experience in further and higher education. He has been published in peer-reviewed journals, including the Journal of Clinical Psychology.
Learn about our Editorial Process
Olivia Guy-Evans, MSc
Associate Editor for Simply Psychology
BSc (Hons) Psychology, MSc Psychology of Education
Olivia Guy-Evans is a writer and associate editor for Simply Psychology. She has previously worked in healthcare and educational sectors.
On This Page:
Case studies are in-depth investigations of a person, group, event, or community. Typically, data is gathered from various sources using several methods (e.g., observations & interviews).
The case study research method originated in clinical medicine (the case history, i.e., the patient’s personal history). In psychology, case studies are often confined to the study of a particular individual.
The information is mainly biographical and relates to events in the individual’s past (i.e., retrospective), as well as to significant events that are currently occurring in his or her everyday life.
The case study is not a research method, but researchers select methods of data collection and analysis that will generate material suitable for case studies.
Freud (1909a, 1909b) conducted very detailed investigations into the private lives of his patients in an attempt to both understand and help them overcome their illnesses.
This makes it clear that the case study is a method that should only be used by a psychologist, therapist, or psychiatrist, i.e., someone with a professional qualification.
There is an ethical issue of competence. Only someone qualified to diagnose and treat a person can conduct a formal case study relating to atypical (i.e., abnormal) behavior or atypical development.
Famous Case Studies
- Anna O – One of the most famous case studies, documenting psychoanalyst Josef Breuer’s treatment of “Anna O” (real name Bertha Pappenheim) for hysteria in the late 1800s using early psychoanalytic theory.
- Little Hans – A child psychoanalysis case study published by Sigmund Freud in 1909 analyzing his five-year-old patient Herbert Graf’s house phobia as related to the Oedipus complex.
- Bruce/Brenda – Gender identity case of the boy (Bruce) whose botched circumcision led psychologist John Money to advise gender reassignment and raise him as a girl (Brenda) in the 1960s.
- Genie Wiley – Linguistics/psychological development case of the victim of extreme isolation abuse who was studied in 1970s California for effects of early language deprivation on acquiring speech later in life.
- Phineas Gage – One of the most famous neuropsychology case studies analyzes personality changes in railroad worker Phineas Gage after an 1848 brain injury involving a tamping iron piercing his skull.
Clinical Case Studies
- Studying the effectiveness of psychotherapy approaches with an individual patient
- Assessing and treating mental illnesses like depression, anxiety disorders, PTSD
- Neuropsychological cases investigating brain injuries or disorders
Child Psychology Case Studies
- Studying psychological development from birth through adolescence
- Cases of learning disabilities, autism spectrum disorders, ADHD
- Effects of trauma, abuse, deprivation on development
Types of Case Studies
- Explanatory case studies : Used to explore causation in order to find underlying principles. Helpful for doing qualitative analysis to explain presumed causal links.
- Exploratory case studies : Used to explore situations where an intervention being evaluated has no clear set of outcomes. It helps define questions and hypotheses for future research.
- Descriptive case studies : Describe an intervention or phenomenon and the real-life context in which it occurred. It is helpful for illustrating certain topics within an evaluation.
- Multiple-case studies : Used to explore differences between cases and replicate findings across cases. Helpful for comparing and contrasting specific cases.
- Intrinsic : Used to gain a better understanding of a particular case. Helpful for capturing the complexity of a single case.
- Collective : Used to explore a general phenomenon using multiple case studies. Helpful for jointly studying a group of cases in order to inquire into the phenomenon.
Where Do You Find Data for a Case Study?
There are several places to find data for a case study. The key is to gather data from multiple sources to get a complete picture of the case and corroborate facts or findings through triangulation of evidence. Most of this information is likely qualitative (i.e., verbal description rather than measurement), but the psychologist might also collect numerical data.
1. Primary sources
- Interviews – Interviewing key people related to the case to get their perspectives and insights. The interview is an extremely effective procedure for obtaining information about an individual, and it may be used to collect comments from the person’s friends, parents, employer, workmates, and others who have a good knowledge of the person, as well as to obtain facts from the person him or herself.
- Observations – Observing behaviors, interactions, processes, etc., related to the case as they unfold in real-time.
- Documents & Records – Reviewing private documents, diaries, public records, correspondence, meeting minutes, etc., relevant to the case.
2. Secondary sources
- News/Media – News coverage of events related to the case study.
- Academic articles – Journal articles, dissertations etc. that discuss the case.
- Government reports – Official data and records related to the case context.
- Books/films – Books, documentaries or films discussing the case.
3. Archival records
Searching historical archives, museum collections and databases to find relevant documents, visual/audio records related to the case history and context.
Public archives like newspapers, organizational records, photographic collections could all include potentially relevant pieces of information to shed light on attitudes, cultural perspectives, common practices and historical contexts related to psychology.
4. Organizational records
Organizational records offer the advantage of often having large datasets collected over time that can reveal or confirm psychological insights.
Of course, privacy and ethical concerns regarding confidential data must be navigated carefully.
However, with proper protocols, organizational records can provide invaluable context and empirical depth to qualitative case studies exploring the intersection of psychology and organizations.
- Organizational/industrial psychology research : Organizational records like employee surveys, turnover/retention data, policies, incident reports etc. may provide insight into topics like job satisfaction, workplace culture and dynamics, leadership issues, employee behaviors etc.
- Clinical psychology : Therapists/hospitals may grant access to anonymized medical records to study aspects like assessments, diagnoses, treatment plans etc. This could shed light on clinical practices.
- School psychology : Studies could utilize anonymized student records like test scores, grades, disciplinary issues, and counseling referrals to study child development, learning barriers, effectiveness of support programs, and more.
How do I Write a Case Study in Psychology?
Follow specified case study guidelines provided by a journal or your psychology tutor. General components of clinical case studies include: background, symptoms, assessments, diagnosis, treatment, and outcomes. Interpreting the information means the researcher decides what to include or leave out. A good case study should always clarify which information is the factual description and which is an inference or the researcher’s opinion.
1. Introduction
- Provide background on the case context and why it is of interest, presenting background information like demographics, relevant history, and presenting problem.
- Compare briefly to similar published cases if applicable. Clearly state the focus/importance of the case.
2. Case Presentation
- Describe the presenting problem in detail, including symptoms, duration,and impact on daily life.
- Include client demographics like age and gender, information about social relationships, and mental health history.
- Describe all physical, emotional, and/or sensory symptoms reported by the client.
- Use patient quotes to describe the initial complaint verbatim. Follow with full-sentence summaries of relevant history details gathered, including key components that led to a working diagnosis.
- Summarize clinical exam results, namely orthopedic/neurological tests, imaging, lab tests, etc. Note actual results rather than subjective conclusions. Provide images if clearly reproducible/anonymized.
- Clearly state the working diagnosis or clinical impression before transitioning to management.
3. Management and Outcome
- Indicate the total duration of care and number of treatments given over what timeframe. Use specific names/descriptions for any therapies/interventions applied.
- Present the results of the intervention,including any quantitative or qualitative data collected.
- For outcomes, utilize visual analog scales for pain, medication usage logs, etc., if possible. Include patient self-reports of improvement/worsening of symptoms. Note the reason for discharge/end of care.

4. Discussion
- Analyze the case, exploring contributing factors, limitations of the study, and connections to existing research.
- Analyze the effectiveness of the intervention,considering factors like participant adherence, limitations of the study, and potential alternative explanations for the results.
- Identify any questions raised in the case analysis and relate insights to established theories and current research if applicable. Avoid definitive claims about physiological explanations.
- Offer clinical implications, and suggest future research directions.
5. Additional Items
- Thank specific assistants for writing support only. No patient acknowledgments.
- References should directly support any key claims or quotes included.
- Use tables/figures/images only if substantially informative. Include permissions and legends/explanatory notes.
- Provides detailed (rich qualitative) information.
- Provides insight for further research.
- Permitting investigation of otherwise impractical (or unethical) situations.
Case studies allow a researcher to investigate a topic in far more detail than might be possible if they were trying to deal with a large number of research participants (nomothetic approach) with the aim of ‘averaging’.
Because of their in-depth, multi-sided approach, case studies often shed light on aspects of human thinking and behavior that would be unethical or impractical to study in other ways.
Research that only looks into the measurable aspects of human behavior is not likely to give us insights into the subjective dimension of experience, which is important to psychoanalytic and humanistic psychologists.
Case studies are often used in exploratory research. They can help us generate new ideas (that might be tested by other methods). They are an important way of illustrating theories and can help show how different aspects of a person’s life are related to each other.
The method is, therefore, important for psychologists who adopt a holistic point of view (i.e., humanistic psychologists ).
Limitations
- Lacking scientific rigor and providing little basis for generalization of results to the wider population.
- Researchers’ own subjective feelings may influence the case study (researcher bias).
- Difficult to replicate.
- Time-consuming and expensive.
- The volume of data, together with the time restrictions in place, impacted the depth of analysis that was possible within the available resources.
Because a case study deals with only one person/event/group, we can never be sure if the case study investigated is representative of the wider body of “similar” instances. This means the conclusions drawn from a particular case may not be transferable to other settings.
Because case studies are based on the analysis of qualitative (i.e., descriptive) data , a lot depends on the psychologist’s interpretation of the information she has acquired.
This means that there is a lot of scope for Anna O , and it could be that the subjective opinions of the psychologist intrude in the assessment of what the data means.
For example, Freud has been criticized for producing case studies in which the information was sometimes distorted to fit particular behavioral theories (e.g., Little Hans ).
This is also true of Money’s interpretation of the Bruce/Brenda case study (Diamond, 1997) when he ignored evidence that went against his theory.
Breuer, J., & Freud, S. (1895). Studies on hysteria . Standard Edition 2: London.
Curtiss, S. (1981). Genie: The case of a modern wild child .
Diamond, M., & Sigmundson, K. (1997). Sex Reassignment at Birth: Long-term Review and Clinical Implications. Archives of Pediatrics & Adolescent Medicine , 151(3), 298-304
Freud, S. (1909a). Analysis of a phobia of a five year old boy. In The Pelican Freud Library (1977), Vol 8, Case Histories 1, pages 169-306
Freud, S. (1909b). Bemerkungen über einen Fall von Zwangsneurose (Der “Rattenmann”). Jb. psychoanal. psychopathol. Forsch ., I, p. 357-421; GW, VII, p. 379-463; Notes upon a case of obsessional neurosis, SE , 10: 151-318.
Harlow J. M. (1848). Passage of an iron rod through the head. Boston Medical and Surgical Journal, 39 , 389–393.
Harlow, J. M. (1868). Recovery from the Passage of an Iron Bar through the Head . Publications of the Massachusetts Medical Society. 2 (3), 327-347.
Money, J., & Ehrhardt, A. A. (1972). Man & Woman, Boy & Girl : The Differentiation and Dimorphism of Gender Identity from Conception to Maturity. Baltimore, Maryland: Johns Hopkins University Press.
Money, J., & Tucker, P. (1975). Sexual signatures: On being a man or a woman.
Further Information
- Case Study Approach
- Case Study Method
- Enhancing the Quality of Case Studies in Health Services Research
- “We do things together” A case study of “couplehood” in dementia
- Using mixed methods for evaluating an integrative approach to cancer care: a case study
Related Articles
Research Methodology
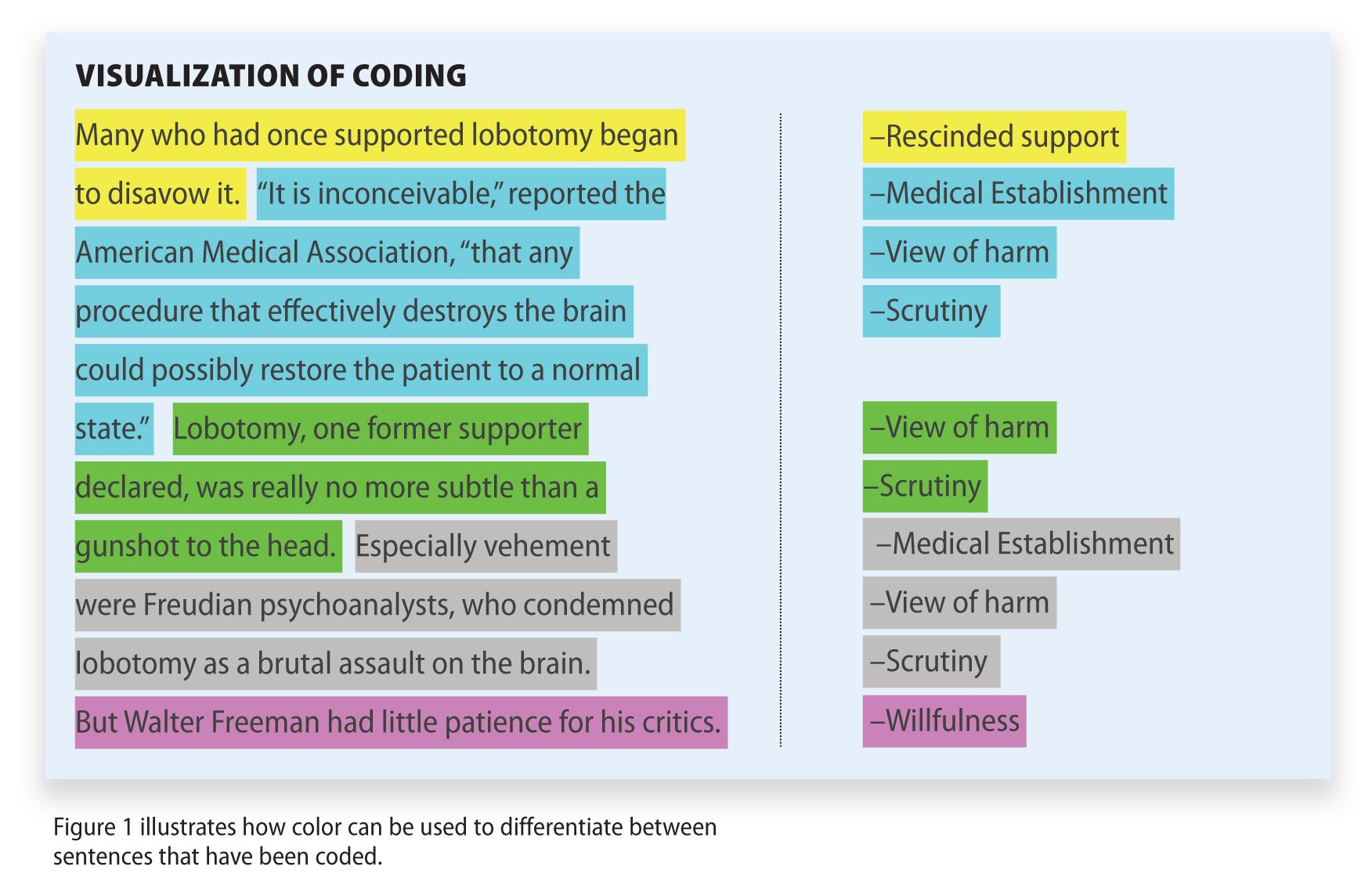
Qualitative Data Coding

What Is a Focus Group?

Cross-Cultural Research Methodology In Psychology

What Is Internal Validity In Research?

Research Methodology , Statistics
What Is Face Validity In Research? Importance & How To Measure

Criterion Validity: Definition & Examples
- Open access
- Published: 06 April 2016
How to review a case report
- Rakesh Garg 1 ,
- Shaheen E. Lakhan 2 &
- Ananda K. Dhanasekaran 3
Journal of Medical Case Reports volume 10 , Article number: 88 ( 2016 ) Cite this article
71k Accesses
19 Citations
3 Altmetric
Metrics details
Peer Review reports
Introduction
Sharing individual patient experiences with clinical colleagues is an essential component of learning from each other. This sharing of information may be made global by reporting in a scientific journal. In medicine, patient management decisions are generally based on the evidence available for use of a particular investigation or technology [ 1 ]. The hierarchical rank of the evidence signifies the probability of bias. The higher up the hierarchy, the better its reliability and thus its clinical acceptance (Table 1 ). Though case reports remain lowest in the hierarchy of evidence, with meta-analysis representing the highest level, they nevertheless constitute important information with regard to rare events and may be considered as anecdotal evidence [ 2 ] (Table 1 ). Case reports may stimulate the generation of new hypotheses, and thus may support the emergence of new research.
The definition of a case report or a case series is not well defined in the literature and has been defined variously by different journals and authors. However, the basic definition of a case report is the detailed report of an individual including aspects like exposure, symptoms, signs, intervention, and outcome. It has been suggested that a report with more than four cases be called a case series and those with fewer than four a case report [ 3 ]. A case series is descriptive in design. Other authors describe “a collection of patients” as a case series and “a few patients” as a case report [ 4 ]. We suggest that should more than one case be reported, it may be defined as a case series—a concept proposed by other authors [ 5 ].
The importance of case reports
A case report may describe an unusual etiology, an unusual or unknown disorder, a challenging differential diagnosis, an unusual setting for care, information that can not be reproduced due to ethical reasons, unusual or puzzling clinical features, improved or unique technical procedures, unusual interactions, rare or novel adverse reactions to care, or new insight into the pathogenesis of disease [ 6 , 7 ]. In recent years, the publication of case reports has been given low priority by many high impact factor journals. However, the need for reporting such events remains. There are some journals dedicated purely to case reports, such as the Journal of Medical Case Reports , emphasizing their importance in modern literature. In the past, isolated case reports have led to significant advancements in patient care. For example, case reports concerning pulmonary hypertension and anorexic agents led to further trials and the identification of the mechanism and risk factors associated with these agents [ 2 , 8 ].
Reporting and publishing requirements
The reporting of cases varies for different journals. The authors need to follow the instructions for the intended publication. Owing to significant variability, it would be difficult to have uniform publication guidelines for case reports. A checklist called the CARE guidelines is useful for authors writing case reports [ 9 , 10 ]. However, it would be universally prudent to include a title, keywords, abstract, introduction, patient information, clinical findings, timeline, diagnostic assessment, therapeutic interventions, follow-up and outcomes, discussion, patient perspective, and informed consent.
Peer review process
The peer review process is an essential part of ethical and scientific writing. Peer review ultimately helps improve articles by providing valuable feedback to the author and helps editors make a decision regarding publication. The peer reviewer should provide unbiased, constructive feedback regarding the manuscript. They may also highlight the strengths and weaknesses of the report. When reviewing an article, it is prudent to read the entire manuscript first to understand the overall content and message. The reviewer than may read section-wise and provide comments to the authors and editorial team accordingly. The reviewer needs to consider the following important points when reviewing a case for possible publication [ 8 , 9 ] (summarized in Table 2 ).
Novelty remains the foremost important aspect of a case. The case report should introduce novel aspects of patient evaluation, investigation, treatment, or any other aspect related to patient care. The relevant information becomes a hypothesis generator for further study. The novelty may at times be balanced with some important information like severe adverse effects, even if they have been reported earlier. Reporting adverse events remains important so that information on cumulative adverse effects can be gathered globally, which helps in preparing a policy or guideline or a warning note for its use in patients. The data related to adverse effects include not only the impact but also the number of patients affected. This becomes more important for serious adverse effects. In the absence of an international registry for adverse effects, published case reports are important pieces of information. Owing to ethical concerns, formal evaluation may not be feasible in the format of prospective study.
Essential description
The case needs to have all essential details to allow a useful conclusion to emerge. For example, if a case is being reported for hemodynamic variability due to a drug, then the drug dose and timing along with timed vital signs need to be described.
Authenticity and genuineness
Honesty remains the most important basic principle of all publications. This remains a primary responsibility of the authors. However, if there is any doubt, reviewers may seek clarification. This doubt may result from some discordance in the case description. At times, a lack of correlation between the figures and description may act as “red flags.” For instance, authors may discuss a technique for dealing with a difficult airway, but the figure is of a normal-appearing airway. Another example would be where the data and figure do not correlate in a hemodynamic response related to a drug or a technique, with the graphical picture or screenshot of hemodynamics acting as an alert sign. Such cause for concern may be communicated in confidence to the editor.
Ethical or competing interests
Ethical issues need to be cautiously interpreted and communicated. The unethical use of a drug or device is not desirable and often unworthy of publication. This may relate to the route or dose of the drug administered. The off-label use of drugs where known side effects are greater than potential benefit needs to be discouraged and remains an example of unethical use. This use may be related to the drug dose, particularly when the drug dose exceeds the routine recommended dose, or to the route of administration. As an example, the maximal dose of acetaminophen (paracetamol) is 4g/day, and if an author reports exceeding this dose, it should be noted why a greater than recommended dose was used. Ultimately, the use of a drug or its route of administration needs to be justified in the manuscript. The reviewers need to serve as content experts regarding the drugs and other technologies used in the case. A literature search by the reviewer provides the data to comment on this aspect.
Competing interests (or conflicts of interest) are concerns that interfere or potentially interfere with presentation, review, or publication. They must be declared by the authors. Conflicts can relate to patient-related professional attributes (like the use of a particular procedure, drug, or instrument) being affected by some secondary gains (financial, non-financial, professional, personal). Financial conflict may be related to ownership, paid consultancy, patents, grants, honoraria, and gifts. Non-financial conflicts may be related to memberships, relationships, appearance as an expert witness, or personal convictions. At times, the conflict may be related to the author’s relationship with an organization or another person. A conflict may influence the interpretation of the outcome in an inappropriate and unscientific manner. Although conflicts may not be totally abolished, they must be disclosed when they reasonably exist. This disclosure should include information such as funding sources, present membership, and patents pending. Reviewers should cautiously interpret any potential bias regarding the outcome of the case based on the reported conflicts. This is essential for transparent reporting of research. At times, competing interests may be discovered by a reviewer and should be included in comments to the editorial team. Such conflicts may again be ascertained when the reviewer reviews the literature during the peer review process. The reviewer should also disclose their own conflicts related to the manuscript review when sending their report to the editorial team.
Impact on clinical practice
This is an important aspect for the final decision of whether to publish a case report. The main thrust or carry-home message needs to be emphasized clearly. It needs to be elaborated upon in concluding remarks.
Patient anonymity, consent, and ethical approval
When reviewing the manuscript of a case report, reviewers should ensure that the patient’s anonymity and confidentiality is protected. The reviewers should check that patient identifiers have been removed or masked from all aspects of the manuscript, whether in writing or within photograph. Identifiers can include things like the name of the patient, geographical location, date of birth, phone numbers, email of the patient, medical record numbers, or biometric identifiers. Utmost care needs to be taken to provide full anonymity for the patient.
Consent is required to participate in research, receive a certain treatment, and publish identifiable details. These consents are for different purposes and need to be explained separately to the patient. A patient’s consent to participate in the research or for use of the drug may not extend to consent for publication. All these aspects of consent must be explained to the patient, written explicitly in the patient’s own language, understood by the patient, and signed by the patient. For the purpose of the case, the patient must understand and consent for any new technique or drug (its dose, route, and timing) being used. In the case of a drug being used for a non-standard indication or route, consent for use must also be described. Patient consent is essential for the publication of a case if patient body parts are displayed in the article. This also includes any identifiers that can reveal the identity of the patient, such as the patient’s hospital identification number, address, and any other unique identifier. In situations where revealing the patient’s identity cannot be fully avoided, for example if the report requires an image of an identifiable body part like the face, then this should be explained to the patient, the image shown to them, and consent taken. Should the patient die, then consent must be obtained from next of kin or legal representative.
With case series, securing individual patient consent is advised and preferable. The authors may also need institutional review board (IRB) approval to publish a case series. IRBs can waive the need for consent if a study is conducted retrospectively and data are collected from patient notes for the purpose of research, usually in an anonymized way. However, wherever possible, individual patient consent is preferable, even for a retrospective study. Consent is mandatory for any prospective data collection for the purpose of publication as a case series. Consent and/or IRB approval must be disclosed in the case report and reasons for not obtaining individual consent may be described, if applicable.
There may be situations in which publishing patient details without their consent is justified, but this is a decision that should be made by the journal editor, who may decide to discuss the case with the Committee on Publication Ethics. Reviewers need to emphasize the issue to the editor when submitting their comments.
Manuscript writing
The CARE guidelines provide a framework that supports transparency and accuracy in the publication of case reports and the reporting of information from patient encounters. The acronym CARE was created from CA (the first two letters in “case”) and RE (the first two letters in “reports”). The initial CARE tools are the CARE checklist and the Case Report Writing Templates. These tools support the writing of case reports and provide data that inform clinical practice guidelines and provide early signals of effectiveness, harms, and cost [ 10 ].
The presentation of the case and its interpretation should be comprehensive and related. The various components of the manuscript should have sufficient information for understanding the key message of the case. The reviewer needs to comment on the relevant components of the manuscript. The reviewer should ascertain that the title of the case manuscript is relevant and includes keywords related to the case. The title should be short, descriptive, and interesting. The abstract should be brief, without any abbreviations, and include keywords. It is preferable to use Medical Subject Headings (MeSH) keywords. Reviewers must ensure that the introduction emphasizes the context of the case and describes the relevance and its importance in a concise and comprehensive manner. The case description should be complete and should follow basic rules of medical communication. The details regarding patient history, physical examination, investigations, differential diagnosis, management, and outcome should be described in chronological order. If repeated observations are present, then they may be tabulated. The use of graphs and figures helps the readers to better understand the case. Interpretation or inferences based on the outcomes should be avoided in this section and should be considered a part of the discussion. The discussion should highlight important aspects of the case, with its interpretation within the context of the available literature. References should be formatted as per the journal style. They should be complete and preferably of recent publications.
Reviewer responsibility
The reviewer’s remarks are essential not only for the editorial team but also for authors. A good peer review requires honesty, sincerity, and punctuality. Even if a manuscript is rejected, the authors should receive learning points from peer review commentary. The best way to review a manuscript is to read the manuscript in full for a gross overview and develop general comments. Thereafter, the reviewer should address each section of the manuscript separately and precisely. This may be done after a literature search if the reviewer needs to substantiate his/her commentary.
Constructive criticism
The reviewer’s remarks should be constructive to help the authors improve the manuscript for further consideration. If the manuscript is rejected, the authors should have a clear indication for the rejection. The remarks may be grouped as major and minor comments. Major comments likely suggest changes to the whole presentation, changing the primary aim of the case report, or adding images. Minor comments may include grammatical errors or getting references for a statement. The editorial team must be able to justify their decision on whether or not to accept an article for publication, often by citing peer review feedback. It is also good style to tabulate a list of the strengths and weaknesses of the manuscript.
Fixed time for review
Reviewer remarks should be submitted within a specified timeframe. If any delay is expected, it should be communicated to the editorial team. Reviewers should not rush to submit feedback without sufficient time to adequately review the paper and perform any necessary literature searches. Should a reviewer be unable to submit the review within the specified timeframe, they should reply to the review invitation to decline at their earliest convenience. If, after accepting a review invitation, the reviewer realizes they do not have time to perform the review, this must be communicated to the editorial team.
Conflict of interest
The reviewer’s conflicts of interest should be included along with the review. The conflicts may be related to the contents of the case, drugs, or devices pertaining to the case; the author(s); or the affiliated institution(s) of the author(s).
Lack of expertise
The reviewer may decline to review the manuscript if they think the topic is out of their area of expertise. If, after accepting an invitation to review, the reviewer realizes they are unable to review the manuscript owing to a lack of expertise in that particular field, they should disclose the fact to the editorial team.
Confidentiality
The reviewer should keep the manuscript confidential and should not use the contents of the unpublished manuscript in any form. Discussing the manuscript among colleagues or any scientific forum or meetings is inappropriate.
Review of revised manuscript
At times, a manuscript is sent for re-review to the reviewer. The reviewer should read the revised manuscript, the author’s response to the previous round of peer review, and the editorial comments. Sometimes, the authors may disagree with the reviewer’s remarks. This issue needs to be elaborated on and communicated with the editor. The reviewer should support their views with appropriate literature references. If the authors justify their reason for disagreeing with the viewer, then their argument should be considered evidence-based. However, if the reviewer still requests the revision, this may be politely communicated to the author and editor with justification for the same. In response to reviewers remarks, authors may not agree fully and provide certain suggestion in the form of clarification related to reviewers remarks. The reviewers should take these clarifications judiciously and comment accordingly with the intent of improving the manuscript further.
Peer reviewers have a significant role in the dissemination of scientific literature. They act as gatekeepers for science before it is released to society. Their sincerity and dedication is paramount to the success of any journal. The reviewers should follow a scientific and justifiable methodology for reviewing a case report for possible publication. Their comments should be constructive for the overall improvement of the manuscript and aid the editorial team in making a decision on publication. We hope this article will help reviewers to perform their important role in the best way possible. We send our best wishes to the reviewer community and, for those who are inspired to become reviewers after reading this article, our warm welcome to the reviewers’ club.
Burns PB, Rohrich RJ, Chung KC. The levels of evidence and their role in evidence-based medicine. Plast Reconstr Surg. 2011;128:305–10.
Article CAS PubMed PubMed Central Google Scholar
Green BN, Johnson CD. How to write a case report for publication. J Chiropr Med. 2006;5:72–82.
Article PubMed PubMed Central Google Scholar
Abu-Zidan FM, Abbas AK, Hefny AF. Clinical “case series”: a concept analysis. Afr Health Sci. 2012;12:557–62.
CAS PubMed PubMed Central Google Scholar
Porta M, editor. A dictionary of epidemiology/edited for the International Epidemiological Association. 5th ed. UK: Oxford University Press; 2008. p. 33.
Medical Research Council of South Africa. Evidence-based medicine. 2016. http://www.mrc.ac.za/healthsystems/sai.htm . Accessed on 1 Nov 2015.
Cohen H. How to write a patient case report. Am J Health-Syst Pharm. 2006;63:1888–92.
Article PubMed Google Scholar
Roberts LW, Coverdale J, Edenharder K, Louie A. How to review a manuscript: a “down-to-earth” approach. Acad Psychiatry. 2004;28:81–7.
Rutowski JL, Cairone JV. How to review scientific manuscripts and clinical case reports for Journal of Oral Implantology. J Oral Implantol. 2009;35:310–4.
Article Google Scholar
Jabs DA. Improving the reporting of clinical case series. Am J Ophthalmol. 2005;139:900–5.
Gagnier JJ, Kienle G, Altman DG, Moher D, Sox H, Riley D; CARE Group. The CARE guidelines: consensus-based clinical case reporting guideline development. BMJ Case Reports. 2013; doi: 10.1136/bcr-2013-201554 .
Download references
Author information
Authors and affiliations.
Department of Anaesthesiology, Pain and Palliative Care, DR BRAIRCH, AIIMS, Ansari Nagar, New Delhi, 110029, India
Rakesh Garg
Neurology and Medical Education, California University of Science and Medicine - School of Medicine, Colton, CA, USA
Shaheen E. Lakhan
Sandwell & West Birmingham Hospitals, NHS Trust, Birmingham, UK
Ananda K. Dhanasekaran
You can also search for this author in PubMed Google Scholar
Corresponding author
Correspondence to Rakesh Garg .
Additional information
Competing interests.
The authors declare that they have no competing interests.
Authors’ contributions
All authors read and approved the final manuscript.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License ( http://creativecommons.org/licenses/by/4.0/ ), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The Creative Commons Public Domain Dedication waiver ( http://creativecommons.org/publicdomain/zero/1.0/ ) applies to the data made available in this article, unless otherwise stated.
Reprints and permissions
About this article
Cite this article.
Garg, R., Lakhan, S.E. & Dhanasekaran, A.K. How to review a case report. J Med Case Reports 10 , 88 (2016). https://doi.org/10.1186/s13256-016-0853-3
Download citation
Received : 27 August 2015
Accepted : 25 February 2016
Published : 06 April 2016
DOI : https://doi.org/10.1186/s13256-016-0853-3
Share this article
Anyone you share the following link with will be able to read this content:
Sorry, a shareable link is not currently available for this article.
Provided by the Springer Nature SharedIt content-sharing initiative
Journal of Medical Case Reports
ISSN: 1752-1947
- Submission enquiries: Access here and click Contact Us
- General enquiries: [email protected]
TechRepublic

8 Best Data Science Tools and Software
Apache Spark and Hadoop, Microsoft Power BI, Jupyter Notebook and Alteryx are among the top data science tools for finding business insights. Compare their features, pros and cons.

EU’s AI Act: Europe’s New Rules for Artificial Intelligence
Europe's AI legislation, adopted March 13, attempts to strike a tricky balance between promoting innovation and protecting citizens' rights.

10 Best Predictive Analytics Tools and Software for 2024
Tableau, TIBCO Data Science, IBM and Sisense are among the best software for predictive analytics. Explore their features, pricing, pros and cons to find the best option for your organization.

Tableau Review: Features, Pricing, Pros and Cons
Tableau has three pricing tiers that cater to all kinds of data teams, with capabilities like accelerators and real-time analytics. And if Tableau doesn’t meet your needs, it has a few alternatives worth noting.

Top 6 Enterprise Data Storage Solutions for 2024
Amazon, IDrive, IBM, Google, NetApp and Wasabi offer some of the top enterprise data storage solutions. Explore their features and benefits, and find the right solution for your organization's needs.
Latest Articles

What is the EU’s AI Office? New Body Formed to Oversee the Rollout of General Purpose Models and AI Act
The AI Office will be responsible for enforcing the rules of the AI Act, ensuring its implementation across Member States, funding AI and robotics innovation and more.

Top Tech Conferences & Events to Add to Your Calendar in 2024
A great way to stay current with the latest technology trends and innovations is by attending conferences. Read and bookmark our 2024 tech events guide.

What is Data Science? Benefits, Techniques and Use Cases
Data science involves extracting valuable insights from complex datasets. While this process can be technically challenging and time-consuming, it can lead to better business decision-making.

Gartner’s 7 Predictions for the Future of Australian & Global Cloud Computing
An explosion in AI computing, a big shift in workloads to the cloud, and difficulties in gaining value from hybrid cloud strategies are among the trends Australian cloud professionals will see to 2028.

OpenAI Adds PwC as Its First Resale Partner for the ChatGPT Enterprise Tier
PwC employees have 100,000 ChatGPT Enterprise seats. Plus, OpenAI forms a new safety and security committee in their quest for more powerful AI, and seals media deals.

What Is Contact Management? Importance, Benefits and Tools
Contact management ensures accurate, organized and accessible information for effective communication and relationship building.

How to Use Tableau: A Step-by-Step Tutorial for Beginners
Learn how to use Tableau with this guide. From creating visualizations to analyzing data, this guide will help you master the essentials of Tableau.

HubSpot CRM vs. Mailchimp (2024): Which Tool Is Right for You?
HubSpot and Mailchimp can do a lot of the same things. In most cases, though, one will likely be a better choice than the other for a given use case.

Top 5 Cloud Trends U.K. Businesses Should Watch in 2024
TechRepublic identified the top five emerging cloud technology trends that businesses in the U.K. should be aware of this year.

Pipedrive vs. monday.com (2024): CRM Comparison
Find out which CRM platform is best for your business by comparing Pipedrive and Monday.com. Learn about their features, pricing and more.

Celoxis: Project Management Software Is Changing Due to Complexity and New Ways of Working
More remote work and a focus on resource planning are two trends driving changes in project management software in APAC and around the globe. Celoxis’ Ratnakar Gore explains how PM vendors are responding to fast-paced change.

SAP vs. Oracle (2024): Which ERP Solution Is Best for You?
Explore the key differences between SAP and Oracle with this in-depth comparison to determine which one is the right choice for your business needs.

How to Create Effective CRM Strategy in 8 Steps
Learn how to create an effective CRM strategy that will help you build stronger customer relationships, improve sales and increase customer satisfaction.

CISOs in Australia Urged to Take a Closer Look at Data Breach Risks
A leading cyber expert in Australia has warned CISOs and other IT leaders their organisations and careers could be at stake if they do not understand data risk and data governance practices.

Snowflake Arctic, a New AI LLM for Enterprise Tasks, is Coming to APAC
Data cloud company Snowflake’s Arctic is promising to provide APAC businesses with a true open source large language model they can use to train their own custom enterprise LLMs and inference more economically.
Create a TechRepublic Account
Get the web's best business technology news, tutorials, reviews, trends, and analysis—in your inbox. Let's start with the basics.
* - indicates required fields
Sign in to TechRepublic
Lost your password? Request a new password
Reset Password
Please enter your email adress. You will receive an email message with instructions on how to reset your password.
Check your email for a password reset link. If you didn't receive an email don't forgot to check your spam folder, otherwise contact support .
Welcome. Tell us a little bit about you.
This will help us provide you with customized content.
Want to receive more TechRepublic news?
You're all set.
Thanks for signing up! Keep an eye out for a confirmation email from our team. To ensure any newsletters you subscribed to hit your inbox, make sure to add [email protected] to your contacts list.

IMAGES
VIDEO
COMMENTS
A case report is a detailed report of the symptoms, signs, diagnosis, treatment, and follow-up of an individual patient. Case reports usually describe an unusual or novel occurrence and as such, remain one of the cornerstones of medical progress and provide many new ideas in medicine. Some reports contain an extensive review of the relevant ...
First steps. Begin by sitting down with your medical team to discuss the interesting aspects of the case and the learning points to highlight. Ideally, a registrar or middle grade will mentor you and give you guidance. Another junior doctor or medical student may also be keen to be involved. Allocate jobs to split the workload, set a deadline ...
Summarize the major points of the case. Provide a limited number (e.g. 3) of takeaway points for the audience. Tailor summary and takeaway points to your audience. Critical Thinking Skills. Successful patient case presentations: Integrate disease and drug knowledge, clinical evidence, and patient factors.
A medical case report, also known as a case study, is a detailed description of a clinical encounter with a patient. The most important aspect of a case report, i.e. the reason you would go to the trouble of writing one, is that the case is sufficiently unique, rare or interesting such that other medical professionals will learn something from it.
Sometimes case reports include a short literature review, if you think it is worthwhile, include it. 2. Describe your patient and follow the diagnostic pathway. For example - Patient X is a 10 ...
Writing a case report is an excellent way of documenting these findings for the wider medical community—sharing new knowledge that will lead to better and safer patient care. For many medical students and junior doctors, a case report may be their first attempt at medical writing. A published case report will look impressive on your ...
5. Complete your introduction and conclusion after you've written the body. Since these sections summarize key points of your case study, it's best to write them last. The introduction gives a brief overview of the basic condition of your patient and the problem you'll address in your case study.
Abstract. Purpose. Guidelines for writing patient case reports, with a focus on medication-related reports, are provided. Summary. The format of a patient case report encompasses the following five sections: an abstract, an introduction and objective that contain a literature review, a description of the case report, a discussion that includes a detailed explanation of the literature review, a ...
Step 2 - Select a Journal. What target audience (doctors & patients) would best benefit from the info? If indexed in PubMed, it is peer reviewed and locatable. Some journals do NOT publish case reports. FIRST double-check the journal's "Author's Guidelines," or "Author's Instructions" to see "Types of Articles".
Patient consent: an ethical requirement for case studies. Informed consent in an ethical requirement for most studies involving humans. It is important to take written consent from the patient before you start writing your case report as all journals will require you to provide patient consent at the time of manuscript submission.
By following these ten steps, you now have a complete set of notes and references for your case report. What you need to do next is put it all together and format it as a case report. This time you will arrange your manuscript differently. Part Two: Writing your second draft Step 11: Start by writing the entire text and listing the references.
Presenting patient cases is a key part of everyday clinical practice. A well delivered presentation has the potential to facilitate patient care and improve efficiency on ward rounds, as well as a means of teaching and assessing clinical competence. 1 The purpose of a case presentation is to communicate your diagnostic reasoning to the listener, so that he or she has a clear picture of the ...
Case study is a research methodology, typically seen in social and life sciences. There is no one definition of case study research.1 However, very simply… 'a case study can be defined as an intensive study about a person, a group of people or a unit, which is aimed to generalize over several units'.1 A case study has also been described as an intensive, systematic investigation of a ...
Case studies are in-depth investigations of a person, group, event, or community. Typically, data is gathered from various sources using several methods (e.g., observations & interviews). The case study research method originated in clinical medicine (the case history, i.e., the patient's personal history). In psychology, case studies are ...
The best way to review a manuscript is to read the manuscript in full for a gross overview and develop general comments. Thereafter, the reviewer should address each section of the manuscript separately and precisely. This may be done after a literature search if the reviewer needs to substantiate his/her commentary.
Case studies to help you to reflect on your practice. These case studies will help you to reflect on your practice, and provide a summary of reflective models that can help aid your reflections and make them more effective. Templates are also provided to guide your own activities. Remember, there is no set way to reflect and you can adapt these ...
CASE 5 CONTINUED NIHSS upon arrival to NICU was 9. The patient experienced a dramatic improvement in symptoms with only mild aphasia and right facial weakness 24 hours post treatment. NIHSS 24 hours post treatment was 2. On hospital day 3, the patient was diagnosed with new onset atrial fibrillation. He was discharged home on hospital day 4 on ...
See 45 CFR 164.501. 3 This guidance is not intended to discuss case reports or presentations for healthcare operations purposes such as education of the covered entity's own workforce or quality improvement purposes. Workforce members presenting individual patient cases for health care operations purposes should be mindful to use or share only ...
Scientific Writer IILocation - Hyderabad #LI Hybrid About the Role:• To write and/or edit under guidance high quality clinical and safety documents: non registration Clinical Study Reports (CSR), Development Safety Update Reports (DSUR), Clinical Trial Registration Documents or Patient Narratives Key Responsibilities:• To write and/or edit under guidance high quality clinical and safety ...
Apache Spark and Hadoop, Microsoft Power BI, Jupyter Notebook and Alteryx are among the top data science tools for finding business insights. Compare their features, pros and cons. By Aminu ...